- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Structural biology
- Structural insights into viral RNA synthesis by influenza polymerase
Structural insights into viral RNA synthesis by influenza polymerase
Influenza virus causes a highly contagious respiratory illness (the ‘flu’) to which everybody on the planet is susceptible. Viruses that infect humans can have three different origins. Seasonal flu causes annual epidemics and has a high public health and economic cost. The estimated global infection rate is 5%–10% in adults and 20%–30% in children and about 250 000 to 500 000 seasonal flu-related deaths occur each year worldwide, mainly amongst the very young, the elderly or the chronically ill. Bird flu is caused by highly pathogenic avian strains such as H5N1 and H7N9 (water birds are the natural reservoir for the virus), which through point mutations, are able to infect humans with up to 60% mortality, but as yet do not transmit between humans. Pandemic flu occurs when novel human strains are generated by recombination between avian and mammalian strains, often via an intermediate host such as the pig. This occurred in 1918, 1957, 1968 and, most recently, in 2009. Pandemics are unpredictable and potentially devastating. While vaccines can provide good protection against influenza, there is a need for effective anti-influenza drugs to treat severe cases and in the event of a dangerous pandemic where no vaccine is available.
The influenza virus genome comprises eight segments of negative sense, single stranded viral RNA (vRNA). Each segment is packaged in an individual ribonucleoprotein particle (RNP). RNPs contain one heterotrimeric RNA-dependent RNA polymerase, which is bound to the conserved 3’ and 5’ extremities of the vRNA (the ‘promoter’) and multiple copies of nucleoprotein. The viral polymerase both transcribes and replicates each genome segment. Transcription, which produces viral mRNA, occurs by a unique cap-snatching mechanism whereby the 5’ cap-structure is cleaved from host cell mRNAs by an endonuclease and stitched onto the beginning of the viral transcript. Replication produces full-length copies of the genomic vRNA and proceeds via a complimentary cRNA intermediate. For many years we have been trying to obtain a high resolution crystal structure of the influenza polymerase in order to understand the distinct mechanisms of RNA synthesis and to enable structure-based drug design targeting this essential viral replication machine.
To overcome the problem of production of sufficient amounts of pure and active recombinant polymerase, we used a novel baculovirus vector, developed by I. Berger (EMBL), in which the three polymerase subunits are expressed as a fusion protein and post-translationally cleaved by a protease into separate pieces. Bat-specific influenza A (FluA) and human influenza B (FluB) polymerases were then co-crystallised with the vRNA promoter and the structures solved at 2.7 Å resolution using data collected at beamline ID23-1. These are the first structures of a complete polymerase from any negative strand RNA virus.
 |
|
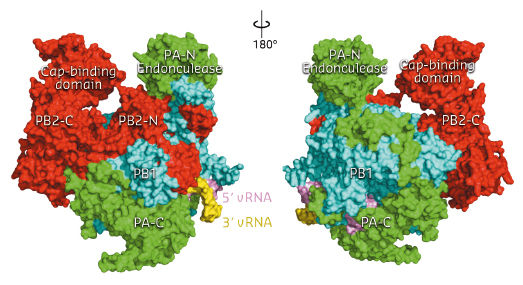
Fig. 94: Front and back views of the complete influenza polymerase with PA shown in green, PB1 in cyan and PB2 in red. The 3’ and 5’ ends of the viral RNA are shown in yellow and violet respectively. |
The three polymerase subunits, PA, PB1 and PB2 mutually stabilise each other through extensive and intricate interfaces (Figure 94). PB1 has a canonical polymerase fold, with a large, enclosed catalytic and RNA binding cavity connected to solvent by channels which allow nucleotide triphosphate (NTP) entrance, template entrance and product exit. The domains involved in cap-snatching, the PA endonuclease domain and the PB2 cap-binding domain, form protrusions which face each other across a solvent channel. Ten nucleotides at the extreme 5’ end of the promoter form a compact stem-loop structure (‘hook’) which is bound in a pocket formed by PB1 and PA. The next four 5’ end nucleotides form canonical base-pairs with the distal part of the promoter 3’ end. The 3’ end proximal nucleotides interact with all three subunits and are directed towards the template entrance to the polymerase active site.
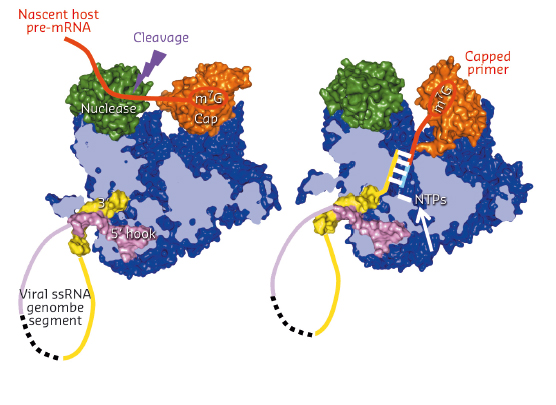
Comparing the FluA and FluB polymerase structures gives mechanistic insight into transcription and replication. In the FluA structure, an ordered priming loop suggests that influenza might initiate unprimed template replication by a similar mechanism to that seen for hepatitis C and Dengue virus polymerases. Interestingly, the FluA and FluB structures differ in the orientation of the PB2 cap-binding domain, suggesting a mechanism for cap-dependent transcription. In the FluA structure, the cap-binding site faces the PA endonuclease active site, a configuration compatible with cap-snatching. In the FluB structure, the cap-binding domain has rotated in situ by 70° and RNA-like residual extra electron density descends part way from the cap binding site towards the PB1 active site. Modelling this as RNA, its prolongation is compatible with the primer strand in known structures of other RNA polymerases bound to primer-template duplexes (Figure 95). The tight binding of the 5’ hook of the viral promoter to the polymerase is compatible with the role of this element in allosterically activating polymerase functions and in stalling transcription at the 5’ proximal oligo-U stretch on the vRNA leading to poly(A)-tail generation on the viral mRNA.
 |
|
Fig. 95: Schematic diagram showing a model of how cap-snatching (left) and cap-dependent priming of transcription (right) works by in situ rotation of the cap-binding domain (orange). |
These structures not only lay the basis for an atomic-level mechanistic understanding of the multiple functions of influenza polymerase but will also shed light on host-specific polymerase variants and open new opportunities for anti-influenza drug design.
Principal publications and authors
A. Pflug, D. Guilligay, S. Reich and S. Cusack, Nature 516, 355–360 (2014); and S. Reich et al., Nature 516, 361–366 (2014).
European Molecular Biology Laboratory, Grenoble Outstation (France)



