- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Electronic structure and magnetism
- Tuning the magnetism of 3d-metal phthalocyanine adlayers by electron doping
Tuning the magnetism of 3d-metal phthalocyanine adlayers by electron doping
Thin phthalocyanine films have been incorporated in various devices including field effect transistors, light-emitting devices and photovoltaic cells. Phthalocyanines can accommodate a variety of metal ions in their centre that have particular spin configurations rendering this class of compounds also interesting for magnetic materials [1]. Due to the particular coordination geometry of the metal ions, the spin states of the complexes are sensitive to their local chemical environment. Bringing the molecules into contact with metal electrodes could therefore have significant influence on the magnetic properties of the metal centres. Another effective route for tailoring the electronic and magnetic characteristics of molecular magnets is offered by direct chemical doping.
The charging sequence of metal-phthalocyanines involves both ligand and metal orbitals and depends strongly on the nature of the central metal ion. Doping of phthalocyanine chemisorbed on a metal substrate is further complicated due to the interplay of additional substrate induced charging and screening effects that can alter the role of the charge donor towards the molecules. The step-wise attachment of electron donor Li atoms to chemisorbed CuPc and NiPc molecules was recently investigated by scanning probe microscopy and DFT calculations [2]. The effects of alkali metal doping on the magnetic properties of metal-phthalocyanines, however, have not been studied in a systematic way, neither in the bulk nor at interfaces. These effects are of particular importance for molecular electronic and spintronic applications.
 |
|
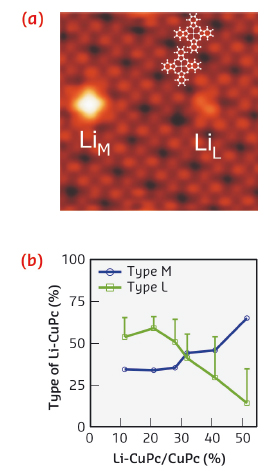
Fig. 13: STM topography of a CuPc adlayer with two doped complexes of different types (8.3 nm x 8.3 nm). Statistical analysis of the type of dopant as a function of Li coverage extracted from the STM data. Error bars account for undefined configurations. |
This study focuses on the effect of Li doping on the electronic and magnetic states of the metal ions for a series of 3d metal-phthalocyanine monolayers (MnPc, FePc, NiPc, CuPc) adsorbed on Ag(001). Our systematic investigation employed X-ray magnetic circular dichroism (XMCD) at beamline ID08 (now ID32) complemented by atomic multiplet calculations, which allowed us to accurately determine changes of the spin, electronic valence, and crystal field in electron-doped phthalocyanines. Figure 13a shows a close-packed array of CuPc. The addition of a small amount of Li atoms leads to two major types of Li-CuPc complexes where the Li atoms can reside at the ligand position (denoted L) or close to the metal centre (denoted M). The relative proportion of each type of complex depends on the Li coverage. With increasing Li coverage, the preference changes from L-type to the M configuration even though there are more ligand sites available than central positions (Figure 13b). The different Li dopant types lead to site specific electron donation either to the metal d-states or to the organic backbone.
 |
|
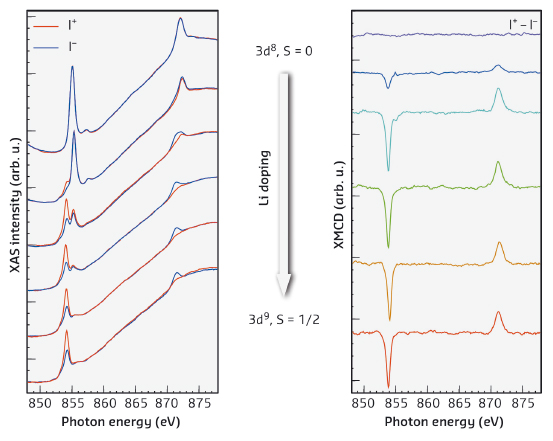
Fig. 14: Evolution of NiPc XAS and XMCD at normal incidence with subsequent Li atom deposition. |
Figure 14 shows the X-ray absorption and XMCD spectra of NiPc as a function of Li doping. With increasing Li concentration, the pristine non-magnetic Ni ions, which have a spin singlet d8 configuration, become reduced and assume a d9 configuration. A magnetic moment corresponding to S = 1/2 appears at the same time, as evidenced by the XMCD. The Li doping has the opposite effect For CuPc: the magnetic moment can be turned off by Li doping, due to the complete filling of the d-shell (from d9 to d10). In contrast, for MnPc, we observed no changes of the d5 metal ion valence state as a function of doping, but a transition from an intermediate S = 3/2 to a high S = 5/2 spin state. This transition is due to a strong reduction of the ligand field induced by Li, which is also observed for CuPc, NiPc, and FePc.
When combined with STM studies (Figure 13 and Ref. [2]), these results provide a complete picture of chemically-doped metal-phthalocyanines on surfaces, in which strong correlation effects induce site-specific charging and magnetic effects. Moreover, these results demonstrate that different spin states can be realised in metal-phthalocyanine layers interfaced with metals by doping with Li atoms, offering a route to tune the magnetic properties of surface-supported molecular systems employing non-magnetic dopants.
Principal publication and authors
S. Stepanow (a), A. Lodi Rizzini (b), C. Krull (b), J. Kavich (b), J.C. Cezar (c), F. Yakhou-Harris (c), P.M. Sheverdyaeva (d), P. Moras (d), C. Carbone (d), G. Ceballos (b), A. Mugarza (b) and P. Gambardella (a,b), J. Am. Chem. Soc. 136, 5451-5459 (2014).
(a) Department of Materials, ETH Zürich (Switzerland)
(b) Catalan Institute of Nanoscience and Nanotechnology (ICN2), Barcelona (Spain)
(c) ESRF
(d) Istituto di Struttura della Materia, CNR, Trieste (Italy)
References
[1] J. Bartolomé, C. Monton and I.K. Schuller, in Molecular Magnets, J. Bartolomé, F. Luis, J.F. Fernández (Eds.), Springer-Verlag, Berlin Heidelberg, 221-245 (2014).
[2] C. Krull, R. Robles, A. Mugarza and P. Gambardella, Nature Mater. 12, 337 (2013).



