- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2013
- Structure of materials
- Fixed-energy X-ray absorption voltammetry
Fixed-energy X-ray absorption voltammetry
To understand the phenomena occurring while a chemical reaction is in progress requires a determination of the oxidation state of a given element, which depends on several factors including its chemical environment and physicochemical conditions. This represents crucial information, whether the element plays the role of reactant/product or of catalyst, since most catalytic cycles involve continuous, periodic transitions between two or more oxidation states of the catalytic centre. This is particularly true in the study of heterogeneous catalysts, where the co-presence of different types of surface sites and the need for a supporting material makes investigation difficult.
X-ray absorption spectroscopy (XAS) represents the tool of choice here since it allows an element selective investigation of the oxidation state and chemical surroundings of a material under “in situ” or “in operando” conditions. Accepting the need for synchrotron radiation, the main limit of XAS is the time required for the recording of spectra and their interpretation. This aspect is partially compensated by QEXAFS (quick scanning EXAFS), allowing the acquisition of spectra within seconds. However, QEXAFS requires a continuous motion of the monochromator, and it is not available at all beamlines. Dispersive XAS can also allow the acquisition of a spectrum in the millisecond scale, but it requires a sophisticated and dedicated beamline.
Here, we introduce fixed-energy X-ray absorption voltammetry (FEXRAV) as a novel rapid XAS technique that can be applied to electrochemical systems for the in situ study of electrode materials. It consists of recording the X-ray absorption coefficient µ at a fixed energy while varying the electrode potential according to a predefined time function. The most common example is a triangular-shaped profile (cyclic voltammetry, CV), but any other shape is in principle applicable. The energy is chosen on the absorption edge in order to give the maximum contrast between different oxidation states of a given element. It follows that any shift from the original oxidation state determines a variation of the absorption coefficient.
Here we show that FEXRAV allows the variation of the oxidation states of the element under study within a selected potential window to be rapidly determined. With this aim, we used highly-hydrated iridium oxide films.
IrOx has been studied for about four decades as one of the most active electrocatalysts for water oxidation, which is the limiting reaction in electrochemical or photoelectrochemical processes that lead to the production of hydrogen, i.e. for effective extraction of energy from renewable sources.
 |
|
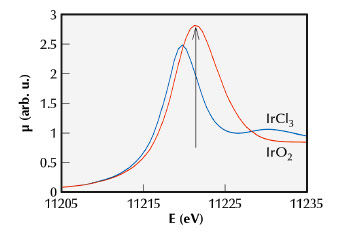
Fig. 132: Normalised XANES spectra of IrO2 (red) and IrCl3 (blue). The arrow marks the energy chosen for the FEXRAV measurements (11221.0 eV) reported in Figure 133. |
Figure 132 describes the basic idea of FEXRAV. The XAS spectrum at the Ir-LIII edge of IrO2 (used as a standard for Ir4+) is plotted with that of IrCl3 (used as a standard for Ir3+). It is evident that fixing the energy at the maximum of the absorption coefficient for IrO2 (at 11221.0 eV, as indicated by the arrow) gives the maximum contrast between Ir3+ and Ir4+.
 |
|
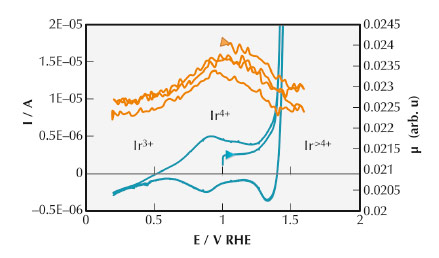
Fig. 133: Conventional cyclic voltammetry (blue line) and the relevant FEXRAV curve (red lines) of iridium oxide recorded at 1 mV s-1. |
With the X-ray energy fixed at 11211.0 eV, the FEXRAV curve plotted in Figure 133 shows that the mean oxidation of Ir state crosses at least three different values while the applied potential was varied. The maximum of µ is in fact located at about 1 V (RHE) that corresponds to Ir4+.
At lower potentials, the decrease in µ indicates the change of oxidation state toward a lower value, i.e. Ir3+.At larger potentials, i.e. for E >1.0 V, µ decreases with increasing Ir average charge state. This last evidence is strictly related to the role of IrOx as electrocatalyst for water oxidation [1].
In conclusion, FEXRAV allows us to rapidly study any species that can be immobilised onto a conductive substrate, in terms of its oxidation state (or any other property that causes a change in the X-ray absorption coefficient) while being dependent on the applied potential. We can screen samples for the application of more detailed local structural probes (XANES, EXAFS), rapidly define the potential windows at which each oxidation state is prevalent, extract important information on the reaction mechanisms, and extract kinetic constants or follow the reaction progression. Also, the latter case is useful especially considering that the FEXRAV signal is not influenced by “parasitic” electrochemical phenomena.
Principal publication and authors
A. Minguzzi (a), O. Lugaresi (a), C. Locatelli (a), S. Rondinini (a), F. D’Acapito (b), E. Achilli (c) and P. Ghigna (c), Anal. Chem. 85, 7009–7013 (2013).
(a) Dipartimento di Chimica, Università degli Studi di Milano, Milan (Italy)
(b) CNR-IOM-OGG c/o ESRF-GILDA, Grenoble (France)
(c) Dipartimento di Chimica, Università di Pavia, Pavia (Italy)
References
[1] A. Minguzzi, O. Lugaresi, E. Achilli, C. Locatelli, A. Vertova, P. Ghigna and S. Rondinini, submitted.



