- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2013
- Soft condensed matter
- The dynamics of nanorod self-assembly at the liquid/air interface studied with GISAXS
The dynamics of nanorod self-assembly at the liquid/air interface studied with GISAXS
The ability to manipulate the chemical and physical properties of colloidal nanocrystals at the atomic level, together with the ability to assemble them into ordered 2D membranes and 3D solids is currently one of the most fascinating and challenging directions of nanoscience [1]. In particular, direct formation of nanocrystal membranes at the liquid-air interface is emerging as a promising method to fabricate functional ultra-thin films [2]. Previous research has mainly focused on characterisation of the final structures formed after self-assembly of the nanocrystal building blocks. However, in order to achieve a high degree of control over the ordering of the final superstructure, it is pivotal to understand the dynamics and mechanism of the self-assembly process.
Using grazing-incidence small-angle X-ray scattering (GISAXS) we have studied dynamics of self-assembly of CdS/CdSe core/shell nanorods (NRs). These NRs have promising optical and electronic properties, as well as high (photo)chemical stability. Self-assembly by controlled solvent evaporation is known to result in membranes in which the NRs have both positional and orientation order over micrometres. Such membranes have potential applications as active layers in lasers, LEDs and solar cells.
We used NRs with diameters of 4 nm and lengths from 15 to 50 nm. In a first set of experiments, we used ex situ transmission electron microscopy (TEM) on the finished self-assembled structures to show that the final orientation of the NRs can be tuned by changing the NR length and/or the initial NR concentration in the dispersion. More specifically, short NRs (< 20 nm) or a low concentration of the initial NR dispersion results in self-assembled structures with the NRs lying flat at the liquid/air interface (Figure 82a). In contrast, longer NRs (> 20 nm) or high initial NR concentrations yield structures with the NRs standing up (Figure 82b).
 |
|
Fig. 82: Bright-field TEM images of the final self-assembled structures for the case of: (a) short NRs and (b) long NRs. |
To investigate the dynamics of NR self-assembly and understand the tunability of the final NR orientation, we have performed in situ GISAXS experiments at beamline ID10. During evaporation of the NR dispersion, an X-ray beam at grazing incidence probes the toluene/air interface at which the NRs self-assemble. We developed an experimental cell with the NR dispersion in toluene on top of an immiscible liquid layer of diethylene glycol (DEG). This allowed us to keep the toluene/air at constant height with respect to the X-ray beam while the NR dispersion evaporated, by continuously injecting additional DEG into the cell. In this way, we achieved a time resolution of a minute, sufficient to follow the complete process of self-assembly, which takes roughly 30 minutes.
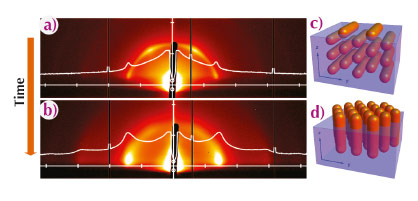
Our study shows first of all that NR self-assembly occurs at the liquid/air interface. Long NRs form ordered structures within minutes that later orient at the liquid/air interface, giving rise to in-plane scattering at q-values relating as 1:√3:2, which is characteristic of 2D hexagonal ordering of NRs standing upright. Short NRs do not show order until after 20 minutes. The final structure shows out-of-plane scattering characteristic of a hexagonal ordering of NRs lying down. Our most striking finding is for NRs of medium length, where we monitored the transition from an intermediate state (Figure 83a), in which the NRs are hexagonally ordered lying down at the liquid/air interface (Figure 83c), to the final superstructure (Figure 83b) which consists of hexagonally ordered upstanding NRs (Figure 83d).
 |
|
Fig. 83: GISAXS patterns of superstructures of medium-length NRs at different times of solvent evaporation: (a) 12 min and (b) 16 min. (c, d) Schematics of the NRs structures (c) at the intermediate stage, and (d) at the end of the self-assembly process. |
Based on the real-time GISAXS data, we developed a model of hierarchical self-organisation that accounts for the dependence of the geometry of the final superstructure on the NR length and the initial NR concentration. According to our model, NRs form ordered bundles already in the bulk dispersion due to mutual van der Waals (vdW) interactions. These bundles are adsorbed at the interface, where they pack together to form a uniform membrane. The orientation of NRs in the final structure depends on the way in which the bundles are adsorbed at the interface, which in turn depends on their size in the bulk dispersion. More precisely, the size of NR bundles increases if the bulk NR concentration increases, or if the NRs are longer, which results in stronger mutual vdW interactions. Our results provide insights into the mechanism of the NR self-assembly at the liquid/air interface, thus paving the way for the exploration of promising applications and sophisticated strategies for the development of functional materials.
Principal publication and authors
F. Pietra (a), F.T. Rabouw (a), W.H. Evers (a,b), D.V. Byelov (c), A.V. Petukhov (c), C. de Mello Donegá (a) and D. Vanmaekelbergh (a), Nano Letters 12, 5515–5523 (2012).
(a) Condensed Matter and Interfaces group, Utrecht University (The Netherlands)
(b) Present address: Department of Chemical Engineering, Delft University of Technology (The Netherlands)
(c) Physical and Colloid Chemistry group, Utrecht University (The Netherlands)
References
[1] D. Vanmaekelbergh, Nano Today, 6, 419-437 (2011).
[2] A. Dong, J. Chen, P.M. Vora, J.M. Kikkawa and C.B. Murray, Nature, 466, 474-477 (2010).



