- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2013
- Structural biology
- Volume-conserving trans-cis isomerisation pathways in photoactive yellow protein visualised by picosecond X-ray crystallography
Volume-conserving trans-cis isomerisation pathways in photoactive yellow protein visualised by picosecond X-ray crystallography
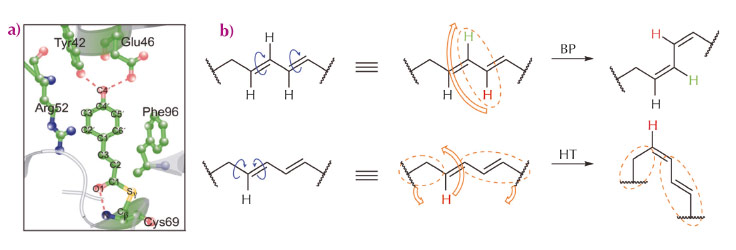
Many photoreceptor proteins convert external light stimuli into molecular action by virtue of isomerisation of its p-conjugated chromophore. Typically, a π-conjugated organic molecule is thought to isomerise via a one-bond-flip mechanism within the gas or solution phases [1]. However, in an inner cavity of a photoreceptor, the chromophore can no longer proceed via one-bond-flip isomerisation, because it interacts with the surrounding side chains of the protein via hydrogen-bonding interactions and steric constraints. In this restrictive condition, isomerisation occurs in a “volume-conserving” fashion. For instance, photoactive yellow protein (PYP) of Halorhodospira halophila has an interior chromophore (p-coumaric acid, pCA, Figure 70a) that is known to be isomerised via the volume-conserved bicycle-pedal (BP) mechanism during its photocycle (Figure 70b) [2]. An alternative, the volume-conserved Hula-Twist (HT) mechanism (Figure 70b), has been observed in various systems, but not in PYP [2]. Both these volume-conserving mechanisms have been observed individually, but these highly-choreographed atomic motions have not been reported in a single system simultaneously.
 |
|
Fig. 70: Isomerisation mechanisms and overview of PYP. a) Close-up of the pCA chromophore and neighbouring residues. b) Schematic description of two volume-conserved isomerisation mechanisms; Bicycle-Pedal (BP), and Hula-Twist (HT). |
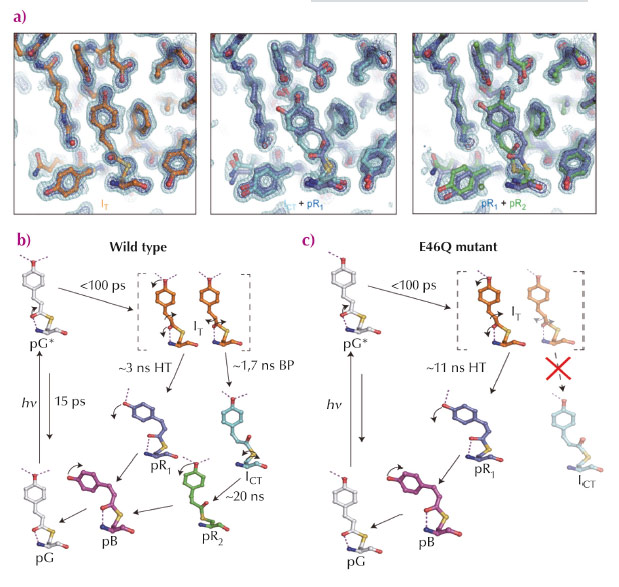
We employed time-resolved Laue crystallography at ESRF beamline ID09TR and APS beamline ID14 to investigate the mechanism of isomerisation of the chromophore of PYP and the structures of intermediate in the pathway from 100 ps to 1 μs. Time-dependent electron density maps depict a highly-twisted structure early on (less than ns) with movement of nearby amino acid residues sustaining hydrogen-bonding networks. Subsequent kinetic analysis unveiled three time-independent electron density maps and two time constants, suggesting that the first map represents homogeneous structure (IT), but the second and third maps have heterogeneous structures (ICT+pR1 and pR1+pR2, respectively) (Figure 71a). Further structure refinement and kinetic analysis revealed four intermediate structures and bifurcated pathways early in the photocycle. In detail, the first intermediate, IT, bifurcates into pR1 (HT pathway) and ICT (BP pathway) with time constants of 3 ns and 1.7 ns, respectively (ICT : pR1 ~ 6:4). ICT further decays into pR2 with a time constant of 20 ns (Figure 71b). Additional experiments with an E46Q mutant indicates that the hydrogen bond network between pCA and nearby residues plays a crucial role in the bifurcation pathways (Figure 71c). Deficiency of one hydrogen bond in the E46Q mutant makes the BP pathway unstable, isomerisation in the E46Q mutant occurs only via the HT pathway. Intensive density functional theory calculations also confirmed that a highly distorted intermediate, IT, is stabilised as an intermediate rather than a transition state as a result of hydrogen bond formation with nearby residues.
 |
|
Fig. 71: a) Time-independent intermediates for WT-PYP recovered from kinetic analysis of time-dependent difference electron density maps. b, c) A photocycle consistent with time-resolved electron density maps of WT-PYP (b) and the E46Q mutant (c). The pathway from IT to ICT via bicycle-pedal mechanism is blocked due to the weaker hydrogen bond between pCA and Q46. |
Our results provide several insights, both structurally and kinetically, into the volume-conserving isomerisation in the PYP photocycle. First, we unveiled a long-hypothesised, highly-distorted intermediate along the trans- to cis- isomerisation pathway. This structure provides consistent and appropriate corroboration of previous experimental and theoretical results [3,4]. Second, the detailed intermediate structures and dynamics revealed that the hydrogen bonds inside proteins play key roles in photo-isomerisation reactions allowing stabilisation of intermediates and a bifurcated pathway. This bifurcated volume-conserving pathway in a single system was found for the first time. Finally, we controlled the reaction pathway just by modifying one hydrogen bond. This study has potential applications not only to other photoactive protein systems but also to time-resolved experiments performed in next generation synchrotrons with femtosecond time-resolution.
Principal publication and authors
Y.O. Jung (a,b), J.H. Lee (b), J. Kim (b), M. Schmidt (c), K. Moffat (d,e), V. rajer (e) and H. Ihee (a,b), Nature Chem. 5, 212220 (2013).
(a) Center for Nanomaterials and Chemical Reactions, Institute for Basic Science (Republic of Korea)
(b) Center for Time-Resolved Diffraction, Department of Chemistry, KAIST (Republic of Korea)
(c) Physics Department, University of Wisconsin–Milwaukee (USA)
(d) Department of Biochemistry and Molecular Biology, and Institute for Biophysical Dynamics, The University of Chicago (USA)
(e) Center for Advanced Radiation Sources, The University of Chicago (USA)
References
[1] R.S. Liu and A.E. Asato, Proc. Natl. Acad. Sci. U.S.A. 82, 259–263 (1985).
[2] Y. Imamoto, M. Kataoka and R.S.H. Liu, Photochem. Photobiol. 76, 584–589 (2007).
[3] L.J.G.W. van Wilderen, M.A. van der Horst, I.H.M. van Stokkum, K.J. Hellingwerf, R. van Grondelle and M.L. Groot, Proc. Natl. Acad. Sci. U.S.A. 103, 15050–15055 (2006).
[4] G. Groenhof, M. Bouxin-Cademartory, B. Hess, S.P. De Visser, H.J.C. Berendsen and M. Olivucci, et al., J. Am. Chem. Soc. 126, 4228–4233 (2004).



