- Home
- Users & Science
- Find a beamline
- Complex systems and biomedical sciences
- ID17 - Biomedical Beamline
- Coronary Angiography
Coronary Angiography
Coronary Angiography at the ESRF
1. Introduction
The program addresses the fundamental concerns associated with conventional angiography, which requires the injection of a contrast agent directely into the coronary arteries by the mean of arterial cathetherization. The potential risks of patient mortality and morbidity, the discomfort and the frequent hospitalizations required (Rogers 1998) prevent its use for routine screening or research. Other techniques as nuclear magnetic resonance angiography (Woodard et al., 1998) or electron beam Computed Tomography (Achenbach et al., 1998) are promising, but despite an active research in these domains, the conventional coronary angiography remains the gold standard technique. At the ESRF, a medical research facility has been commissioned for basic research studies in areas of fundamental bio-medical and pre-clinical research (Elleaume et al 1999). Designed for programs in transvenous coronary angiography, cerebral computed tomography (Dilmanian et al. 1997) and radiotherapy (Laissue et al. 1998), the facility now hosts programs in mammography, bronchography and radiation cell biology. The commissioning of the human angiography program brings to an end the construction phase of the facility. The x-ray energies and intensity are those required for the dual-energy digital subtraction technique to enhance the image contrast of the coronary arteries following venous injection of an iodinated contrast agent. The dilution of the contrast agent prior to arrival in the coronary arteries, the background in the ventricles and the pulmonary veins, and the motion of the heart all present problems which are effectively solved by this technique.
2 Instrumentation
The facility consists of the x-ray beamline that delivers the high intensity x-ray beam from the storage ring source, associated optics and imaging detectors, a patient imaging room, and a patient catheterization laboratory. The ESRF operates at an electron energy of 6 GeV. The x-rays available in the medical facility radiate from a wiggler magnet and are about 4 orders of magnitude more intense than from a conventional source.
The ESRF monochromator is a single cylindrically bent silicon (111) crystal mounted in Laue geometry, which focuses the beam vertically. Two monochromatic beams are produced, one with an energy above and the other below the K-absorption edge of iodine at 33.17 KeV. The total energy bandwidth reflected by the bent Laue crystal is 400 eV. Each beam has an energy bandwidth of 125 eV and their mean energies are separated by 300 eV. The data acquisition system is made of a high purity germanium detector associated with high dynamic range electronics (16 bits). The detector is electrically segmented into 864 strips distributed over two rows, resulting in a detection element pitch of 350 microns. The germanium crystal is 2 mm thick, providing an efficiency of nearly 100% at 33 keV. Since the x-ray beam is fan-shaped (0.7 mm height and 150 mm width) it is necessary to move the patient through the beam in order to obtain two-dimensional angiograms. The positioning system, which allows both for positioning of the patient and the scan motions during the data acquisition, is a high precision stage with seven degrees of freedom. The speed is constant over a length of 200 mm, for a total displacement of 600 mm including the acceleration and deceleration phases. The vertical speed was set at 250 mm/s with an integration time of 1.4 ms corresponding to a vertical displacement of 350 microns per line. The image spatial resolution is therefore 350 m x 350 m and the temporal resolution is one image every 1.3 second. The x-ray dose received by the patient was monitored all along the experiment, it amounted 30 mGy/image in average. The high dose rate available in the monochromatic beam, about 10 Gy/s at 33.17 keV, could represent a real danger to the patient in case of failure, a safety system has been designed to prevent exposition of the patient in such case. The x-ray dose given to the patient is measured with a wide ion chamber, which detects both monochromatic fan-shaped beams at the same time. Two similar dose monitors have been installed to provide the redundancy necessary for safety reasons. They are the last beamline components before the patient. Two independent fast-acting shutters are used in case of emergency to stop the beam, each one is capable of stopping the full monochromatic beam. If a problem is detected by the patient safety system, the x-ray beam is shutdown in 10 ms.
3 Medical Protocol
A research protocol has been designed to evaluate the potential of this method, in comparison with the conventional gold-standard technique. Patients included in the protocol have previously undergone angioplasty. In 30% of the cases (Van’t Hof 1998) re-stenosis occurs after angioplasty. If a re-stenosis is suspected, the patient is imaged at the ESRF in the left anterior oblique (LAO) orientation, and within the next few days with the conventional technique at the hospital. The total radiation dose to a patient during the synchrotron imaging sequence is limited to 0.2 Gy by the medical protocol. Thirty patients are included in this validation phase, which is expected to last one year. The protocol is focused on the right coronary artery in the initial phase, but investigations on the left oblique orientation will also be performed to assess left coronary artery visibility.
4 Imaging Procedure
The catheter is inserted into the brachial vein and advanced to the superior vena cava under fluoroscopy control, the patient is then installed in scanning system in the imaging room. The correct orientation of the patient is first checked by taking a single image at low x-ray dose and without injection of a contrast agent. The transit time between the injection of the contrast agent and the arrival of the bolus in the heart is then measured using a series of five synchrotron images at low x-ray dose (5 mGy) and with a small amount of contrast agent (10 ml). Forty-five minutes later, once the contrast agent used for the transit time estimation has disappeared totally from the venous circulation, the imaging sequence takes place. 30 to 45 ml of iodine (Iomeron 350 mg/ml Bracco Italy) are injected into the superior vena cava using an auto-injector under remote control (15 ml/s). The image sequence is started a few seconds after the injection of the contrast agent depending on the transit time evaluation. Five images are then acquired to follow the iodine bolus through the patient circulation while the positioning system is moving up and down. The time delay between two images is 1.2 second.
5 Results
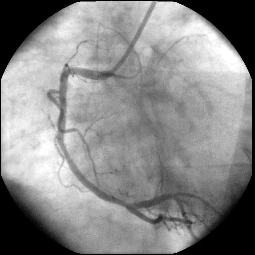
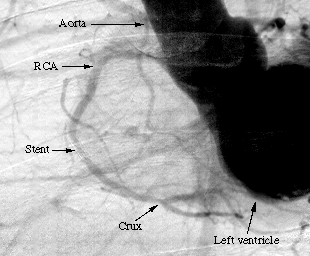
For the first patients, we obtained a good agreement in terms of stenosis diagnosis between synchrotron angiography and the usual angiography procedure in hospital. One image obtained with the first patient at the ESRF is shown on Figure 1a. Stenosis appears visible inside the stent in the second segment C2, and the known distal stenosis visible at the crux remains mild (inferior to 50%). We stress the excellent visualization of the distal part of the right coronary artery (RCA). These findings were in excellent agreement with the conventional selective coronarography perfomed few hours later in the hospital cardiological unit (Figure 1b).
For the last patient investigation, the injection was performed using a short catheter (10 cm long) and excellent image quality was also obtained. This injection mode will be used, when possible, for future examinations.
In conclusion, the synchrotron method provides a good way to assess the status of known stenoses with a less invasive method compared to conventional coronary angiography. Since the method uses venous catheterization, the coronary arteries are not artificially pressurized. The resulting images are therefore in a true physiological state. The virtual absence of complications with this method allows it to by used for research protocols where conventional angiography may not be allowed. In patients with stents, the synchrotron method allows simultaneous visualization of the stent and the perfusion of the vessel. Surgical bypasses are very easy to visualize as well since the catheter does not have to be introduced into the artery. The disadvantages of the synchrotron method are mainly the possible superposition of the venous structures over the arterial tree, and the filling of the ventricles with the contrast agent. These inconveniences can be overcome by carefully selecting the projection views. The left coronary arteries will be investigated within the framework of another medical protocol. As shown by the German studies (Dill 1998), it is possible to vizualize correctly the other coronary segments.
The synchrotron method is a very efficient minimally-invasive techniques to visualize and quantify coronary stenosis. The ESRF has advantages over other synchrotron facilites for this type of research. The beamline has been designed to allow imaging at the gadolinium K-absorption edge in the future. Research with a Gd contrast agent may lead to superior images with lower radiation dose to the patients, particularly for large patients. One of the major strengths of the ESRF program is the close collaboration and support by the staff from the Centre Hopitalier Universitaire. This is essential to the long term development and utilization of the facility.
Acknowlegments: We warmly thank M. Perez, G. Goujon, A. Draperi and Y. Dabin for engineering support, and S Michelland and E Pierrard for patient care during the imaging procedure at the ESRF. Figures captions
Figure 1:
a: Intravenous synchrotron angiogram of the first patient at the ESRF taken in a left anterior oblique (LAO) projection. The image shows the second segment C2 with a stent and the crux. RCA: right coronary artery.
b: Conventional selective coronary angiography of the same patient, in the LAO orientation, performed the same day at the cardiological unit of the hospital after arterial catheterization.
REFERENCES
1 Achenbach S, Moshage W, Ropers D, Bachmann K 1998 Curved multiplanar reconstructions for the evaluation of contrast-enhanced electron beam CT of the coronary arteries. Am. J. Roentgenol. 170 895-899
2 Dill T, Dix WR, Hamm CW, Jung M, Kupper W, Lohman M, Reime B, and Ventura R 1998 Intravenous coronary angiography with synchrotron radiation Eur. J. Phys. 19 499-511
3 Dilmanian FA, Wu XY, Parsons EC, Ren B, Kress J, Button TM, Chapman LD, Coderre JA, Giron F, Greenberg D, Krus DJ, Liang Z, Marcovivi S, Petersen MJ, Roque CT, Shleifer M, Slatkin DN, Thomlinson WC, Yamamoto K and Zhong Z, 1997 Single and dual energy CT with monochromatic synchrotron x-rays. Phys. Med. Biol. 42 371-387
4 Dix WR 1995 Intravenous coronary angiography with synchrotron radiation Prog. Biophys. Molec. Biol. 63 159-191
5 Elleaume H, Charvet AM, Berkvens P, Berruyer G, Brochard T, Dabin Y, Dominguez MC, Draperi A, Fiedler S, Goujon G, Le Duc G, Mattenet M, Nemoz C, Perez M, Renier M, Schulze C, Spanne P, Suortti P, Thomlinson W, Estève F, Bertrand B and Le Bas JF 1999. Instrumentation of the ESRF medical imaging facility Nucl. Instr. Methods. A428 513-527
6 Laissue JA, Geiser G, Spanne PO, Dilmanian FA, Gebbers JO, Geise M, Wu XY, Makar MS, Micca PL, Nawrocky MM, Joel DD Slatkin DN 1998 Neuropathology of ablation of rat gliosarcomas and contiguous brain tissues using a microplanar beam of synchrotron-wiggler-generated x rays Int. J. Cancer 78 654-660
7 Lewis R. 1997 Topical review on Medical applications of synchrotron radiation x-rays. Phys. Med. Biol., 42 1213-1243
8 Ohtsuka S, Sugishita Y, Takeda T, Itai Y, Tada J, and Hyodo K, 1999 Dynamic intravenous coronary angiography using 2D monochromatic synchrotron radiation Br. J. Radiol. 72 24-28
9 Rogers LF 1998, The heart of the matter: non-invasive coronary artery imaging Am. J. Roentgenol. 841 107
10 Rubenstein E, Hofstadter R, Zeman HD, Thompson AC, Otis JN, Brown G, Giacomini JC, Gordon H, Kernoff RS, Harrison DC, and Thomlinson WC 1986 Transvenous coronary angiography in humans using synchrotron radiation Proc. Natl. Acad. Sci. USA 83 9724-9728
11 Thomlinson W 1995 Transvenous Coronary Angiography in Humans Biomedical Applications of Synchrotron Radiation (E Burattini and A Balerna IOS Press Amsterdam) 127-153
12 Van’t Hof AW, De Boer MJ, Suryapranata H, Hoorntje JC, Zijlstra F 1998 Incidence and predictors of restenosis after successful primary coronary angioplasty for acute myocardial infarction: the importance of age and procedural result Am. Heart J. 136:3 518-27
13 Woodard PK, Debiao Li. D, Haacke EM, Dhawale PJ, Kaushikkar S, Barzilai B, Braverman AC, Ludbrook PA, Weiss AN, Brown J J, Mirowitz SA, Pilgram TK, Gutierrez F 1998 Detection of coronary stenoses on source and projection images using three-dimensional MR angiography with retrospective respiratory gating: preliminary experiments. Am. J. Roentgenol. 170 883-888



