- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Electronic structure, magnetism and dynamics
- Operando X-ray absorption spectroscopy shows structural dynamics of Cu-containing catalysts for bio-based butadiene formation
Operando X-ray absorption spectroscopy shows structural dynamics of Cu-containing catalysts for bio-based butadiene formation
The production of commodity chemicals from renewable resources such as biomass, will aid the necessary transition to a more sustainable society. Butadiene, a key building block for rubbers and polymers, such as ABS, can, for instance, be produced from bioethanol. Detailed insight was obtained by operando X-ray absorption spectroscopy into the complex structure of the catalyst materials used for this process, allowing for improved catalyst design.
Future biorefinery operations should produce high-value bio-based chemicals in addition to biofuels in order to be economically competitive. The bio-based chemicals can be either new compounds or can be ‘drop-in’ replacements, i.e. molecularly-identical to the chemical building blocks that we obtain today from petrochemical refining. The latter class holds the advantage of serving existing markets and can make use of existing infrastructure for further processing. One particularly interesting route to a drop-in commodity chemical is the conversion of bio-based ethanol to butadiene [1]. Butadiene, mainly produced as byproduct of naphtha steam cracking, is one of the major chemical commodities with a forecasted production of 12 Mt in 2015 and is used mainly as a monomer for rubbers, polymers and latexes. The one-step conversion of ethanol to butadiene is known as the Lebedev process and has been operated commercially during the first half of the twentieth century in the Soviet Union and elsewhere, eventually losing out to petrochemical production processes. The current availability of large volumes of bioethanol and its possible exploitation as a renewable feedstock has rekindled the academic and industrial interest in this process. SiO2-MgO based catalysts have long been considered as the prototypical catalysts for the Lebedev process and have been applied industrially. The performance of such catalysts can be greatly enhanced if a third component, a transition metal or metal oxide (e.g., metallic Ag or Cr, Ni, Zn, or Cu oxides), is added [2]. Fundamental understanding of the structure-function relationships that govern the performance of these (promoted) materials, together with how they are influenced by the method of catalyst preparation, is required for the efficiency improvements necessary for making this route a viable alternative. However, the knowledge available on the structural and morphological properties of such promoted SiO2-MgO based catalysts, as well as on how the structure evolves under reaction conditions, is still rather limited.
 |
|
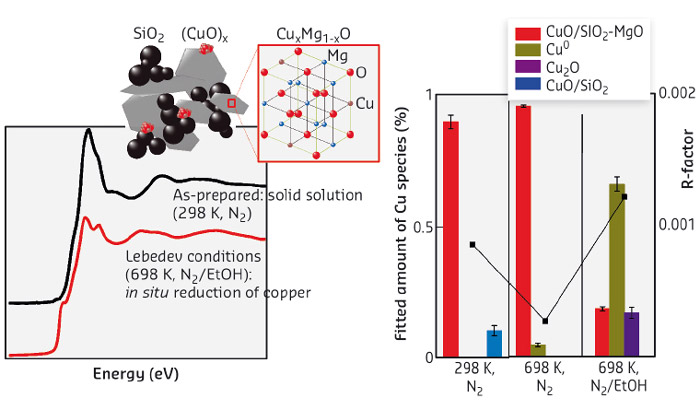
Fig. 71: Left: Operando XAS studies show changes in copper speciation under reaction conditions; right: fitting of the XANES region allowed estimation of the different copper species present under reaction conditions. |
We have studied CuO-promoted SiO2-MgO catalysts in great detail with a combination of characterisation techniques in order to get more insight into the nature, dispersion and role of CuO in the Lebedev process (Figure 71). Operando studies of these Lebedev catalysts also provided insight into the evolution in copper speciation under reaction conditions. Ex situ and operando XANES and EXAFS measurements recorded at beamline BM26A (DUBBLE CRG), provided pivotal information of the speciation, oxidation state and geometry of the Cu promoter in the fresh catalysts and allowed for copper structural changes to be elucidated under typical Lebedev conditions, i.e. in the presence of ethanol at 698 K. As expected, the preparation method of the CuO/SiO2-MgO catalyst strongly influenced its catalytic performance. In combination with other characterisation techniques, ex situ XAS and EXAFS allowed the copper oxidation state and geometry to be firmly established, with all copper found to be Cu2+ in a distorted octahedral geometry in a CuxMg1–xO solid solution. XAS measurements of the CuO/SiO2-MgO catalyst under operando conditions, evidenced by butadiene formation detected by mass spectrometry, provided detailed insight into the remarkable changes in copper speciation under the reducing conditions of the process. The system was shown to evolve to a mixture of copper species, which could be distinguished by fitting the XANES, with 60% of the copper being reduced to Cu0 and the remainder present as Cu+ or Cu2+. This steady state was obtained regardless of the pretreatment history of the catalyst, emphasising the dynamics of the system. Some of the resulting copper species are thought to be involved in boosting the dehydrogenation activity of the catalyst, while others modify the acid-base characteristics of the material, the balance of which is essential to selective butadiene formation. The detailed insights into catalyst structure and its dynamics can now serve as input for the development of the next generation of Lebedev catalysts.
Principal publication and authors
Ex situ and operando studies on the role of copper in Cu-promoted SiO2–MgO catalysts for the Lebedev ethanol-to-butadiene process, C. Angelici (a), F. Meirer (a), A.M.J. van der Eerden (a), H.L. Schaink (a), A. Goryachev (b), J.P. Hofmann (b), E.J.M. Hensen (b), B.M. Weckhuysen (a) and P.C.A. Bruijnincx (a), ACS Catal. 5, 6005-6015 (2015); doi: 10.1021/acscatal.5b00755.
(a) Inorganic Chemistry and Catalysis, Utrecht University (The Netherlands)
(b) Laboratory of Inorganic Materials Chemistry, Eindhoven University of Technology (The Netherlands)
References
[1] C. Angelici et al., ChemSusChem 6, 1595-1614 (2013).
[2] C. Angelici et al., ChemSusChem 7, 2505-2515 (2014).



