- Home
- Industry
- Industry news
- Formulation of a...
Formulation of a urate oxidase enzyme as a crystalline drug
12-05-2010
Microcrystalline suspensions of rasburicase, a urate oxidase enzyme, were characterised using X-ray powder diffraction. The study of the polymorphism of this protein will help identify the best crystalline formulation for subcutaneous administration.
The Challenge
To produce a microcrystalline suspension of rasburicase suitable for use as a subcutaneous drug and to fully characterise the various crystalline products.
Background
Although naturally produced uric acid has beneficial effects in the body, its accumulation can cause diseases including gout. Rasburicase is a recombinant urate oxidase enzyme used as a drug to help the body to eliminate uric acid. It is typically administered to prevent renal failure during the treatment of tumours. The current formulation involves the lyophilised drug in a preparation suitable only for use in hospitals. A crystalline preparation could permit self administration, avoiding a hospital visit. However, to be suitable for administration by injection, the crystals needs to be smaller than 50 micrometres. Full characterisation of the drug’s crystalline polymorphism is required by the health authorities -- this can be carried out by X-ray powder diffraction. There are many added benefits from using a crystalline form of a drug: it permits better control over the release rate into the system by an optimisation of the crystals size and polymorphism, and it should also offer an extended shelf life and a reduction in production costs.
Synchrotron techniques
Ultra high resolution X-ray powder diffraction was used to characterise crystalline suspensions of rasburicase.
Results
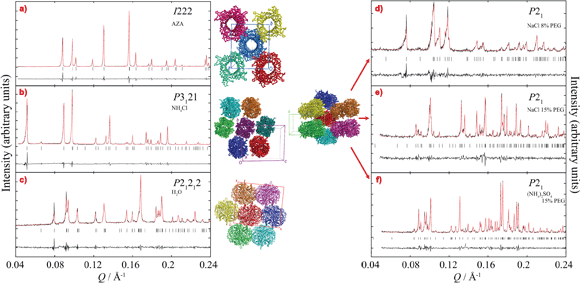
This study demonstrated the feasibility of the production and characterisation of microcrystalline suspensions of the drug. Different polymorphs of rasburicase were obtained through variation of the crystallisation conditions. Each crystalline form was characterised using powder diffraction. A set of conditions could be defined to produce crystals of a suitable size and of known polymorphism. One of the crystallisation conditions in particular involved crystallisation from pure water, avoiding the undesirable addition of chemicals, making it an ideal way of preparing the future drug.
How did the synchrotron experiment help?
Powder diffraction on weakly diffracting samples like proteins is, for the moment, only possible at synchrotron radiations facilities. Powder diffraction permitted identification of the polymorphs of the drug, which could then be associated with known phases (structures already deposited in the PDB), shown in Figure 1.
Further information
Polymorphism of microcrystalline urate oxidase from Aspergillus flavus, I. Collings (a), Y. Watier (a), M. Giffard (b), S. Dagogo (a), R. Kahn (c), F. Bonneté (b), J.P. Wright (a), A.N. Fitch (a) and I. Margiolaki (a,d), Acta Cryst. D66, 539–548 (2010).
(a) ESRF
(b) Centre Interdisciplinaire de Nanoscience de Marseille, CINaM–CNRS UPR 3118, Aix-Marseille Universite, Marseille (France)
(c) Institut de Biologie Structurale, Grenoble (France)
(d) Department of Biology, University of Patras (Greece)
Top image: Microcrystals of urate oxidase.