- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Soft condensed matter
- Watching protein layers grow
Watching protein layers grow
Proteins in an aqueous solution spontaneously form a two-dimensional (2D) layer at the air-water interface. A complex mechanism guides the assembly of proteins through interactions among proteins, interface and the bulk medium. The air-water interface is a convenient medium which provides us with a straightforward way for manipulating these interactions. For instance, decorating a bare water surface with different kinds of surfactant molecules, changing their surface concentration, controlling the temperature and the ionic concentration of the aqueous medium are achieved by simple means. The air-water interface is thus an ideal template for guiding 2D self-assemblies of proteins. Recent years have witnessed the increased use of the air-water interface for growing 2D protein crystals. Inevitably, this has led to a considrable interest in the exploration of mechanism behind self-assembly of proteins at surfaces. However, the hierarchical structures at various length-scales and the associated large range of time-scales pose a big challenge to both experiments and computer simulation.
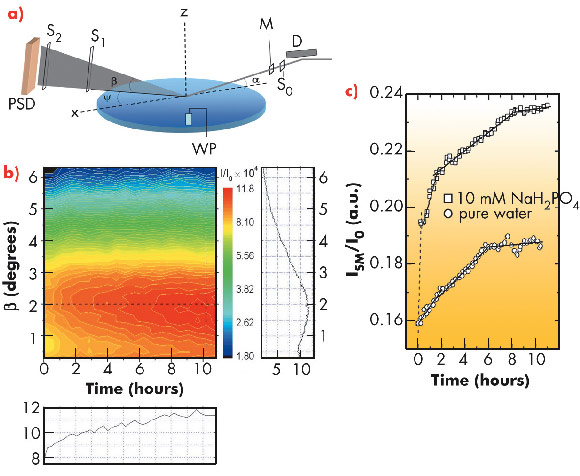
Here we report our experimental study at beamline ID10B on the mechanism of lysozyme monolayer growth at the air-water interface decorated with amphiphilic molecules (ETHT4001). The protein was inserted into the aqueous subphase and the interface was monitored with the grazing incidence X-ray off-specular scattering (GIXOS) technique [1] in a time-resolved experiment keeping the incident angle fixed and measuring the scattering signal on a position sensitive detector as shown in Figure 60a. When the grazing angle is below the critical angle, the scattering signal carries the structural information from an interfacial layer of only ~7 nm thick. Figure 60b demonstrates the evolution of signal on the detector after protein injection into the subphase. The constantly changing signal clearly suggests the dynamic nature of the interface, which systematically evolves towards equilibrium. The integrated signal intensity, Figure 60c, is marked with segmental evolution separated by kinks at various points during the growth.
 |
|
Fig. 60: a) Experimental setup. Deflector (D) tilts the monochromatic X-ray beam to the liquid surface with incidence angle α. The beam size is defined by slit S0. Monitor (M) measures incident intensity I0. Ψ and β are in-plane and out-of-plane scattering angles. Long slits S1 and S2 define the horizontal resolution. PSD - position sensitive detector; WP - Wilhelmy plate. b) The 2D colour map represents the intensity evolution at PSD normalised on monitor. Vertical and horizontal slices of the 2D map corresponding to the dashed lines are on the right and bottom of the map. c) Evolution of intensity integrated on PSD. |
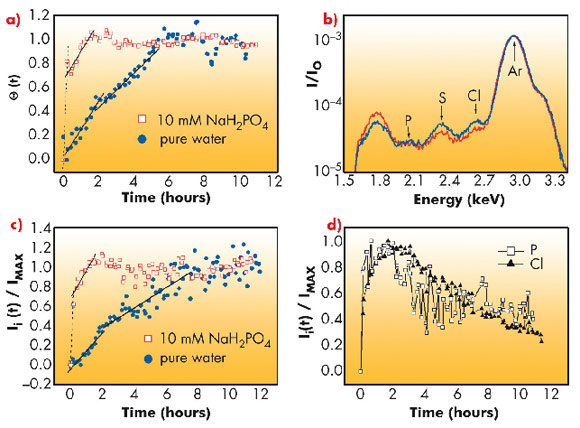
The growth of a protein layer at the interface involves a multitude of processes such as protein adsorption, conformational changes, relaxation via the diffusion of proteins and therefore clustered domains within the interface are expected to occur at different time-scales. The sudden change in growth rate of integrated intensity thus can be understood in terms of a sequence of different dynamical processes. However, the main problem is to make a clear distinction between the intermediate steps. In this study, for the first time, we suggest a scheme to distinguish between adsorption and relaxation processes by simultaneously measuring surface pressure and diffuse X-ray scattering. The obtained protein coverage is shown in Figure 61a. In support of this study we performed another experiment using the grazing incidence X-ray fluorescence (GIXF) technique. In this geometry the fluorescence signal is only sensitive to protein adsorption to the interface and not to the structural relaxation at the interface. The sulphur signal in Figure 61 (b,c) is representative of lysozyme as its polypeptide chain contains 10 sulphur atoms. The counter-ion profiles (Figure 61d) evolve during the entire period of measurement, whereas the sulphur signal attains saturation. The changing counter-ion profile suggests that the protein layer constantly evolves even after protein coverage is saturated. The evolution at this stage is due to the relaxation of the layer through the diffusion of proteins or cluster of proteins along the interface.
 |
|
Fig. 61: a) protein coverage obtained from diffuse X-ray scattering, b) GIXF signal from the interface, c) the evolution of sulphur signal, d) the evolution of counter ions (Cl, P). |
Our study brings new insight into the mechanism of protein monolayer growth at the air-water interface. The time-resolved measurements provide ample evidence to suggest that the growth mechanism involves a sequence of processes characterised with different time-scales. In the hierarchy of evolution, protein adsorption is followed by relaxation via reorganisation of protein at the interface. It was demonstrated here that time-resolved GIXOS and GIXF together brings out crucial information to distinguish between adsorption and relaxation.
Principal publication and authors
A. Singh (a,b), O. Konovalov (a), J. Novak (a,c) and A. Vorobiev (a), Soft Matter 6, 3826 (2010).
(a) ESRF
(b) Current address: Banasthali University, Rajasthan (India)
(c) Current address: Institute of Solid State Physics, Graz (Austria)
References
[1] S. Mora, J. Daillant, D. Luzet and B. Struth, Europhys. Lett., 66, 694 (2004).



