- Home
- Users & Science
- Find a beamline
- Complex systems and biomedical sciences
- ID17 - Biomedical Beamline
- Diffraction-enhanced Imaging
Diffraction-enhanced Imaging
Early detection and characterisation of breast abnormalities like small mass lesions and microcalcifications is crucial to a positive prognosis of breast cancer as they may indicate malignant tumours. The absorption contrast of such signatures in radiographs obtained by standard mammography is limited by the acceptable radiation dose that can be given to the breast. The size of the details that still can be visualised is limited also by dose and by diffuse scattering from the breast tissue.
The DEI technique can reduce considerably these limitations. It can sense phase gradients in the diffraction plane of the analyser crystal that is introduced behind the tissue sample. Since such phase effects can be strong for low Z elements, spectacular contrast enhancements have been achieved in mammograms obtained with this technique [1,2]. In addition, diffuse scattering is intrinsically rejected by the analyser crystal and the possibility for significant dose reduction is offered due to the weaker energy dependence of phase in comparison with absorption [3].
However due to the different nature of the phase dependent contrast process, DEI mammograms differ greatly in appearance and are more complex than conventional images. In order to facilitate the interpretation of DEI images and in particular to identify malignant lesions and differentiate them from normal tissue, we performed a comparison between DEI- and conventional images and the histology of the sample.
A detailed analysis of the samples is based on computed tomography (CT) images taken by the DEI method at slices that are prepared for histo-pathology after X-ray imaging, so that there is an approximate one-to-one correspondence of positions. This also ensures a better identification of details than for planar projection images where different structures in the projection direction can be superimposed. However projection images are used as reference material to make a connection to conventional mammography.
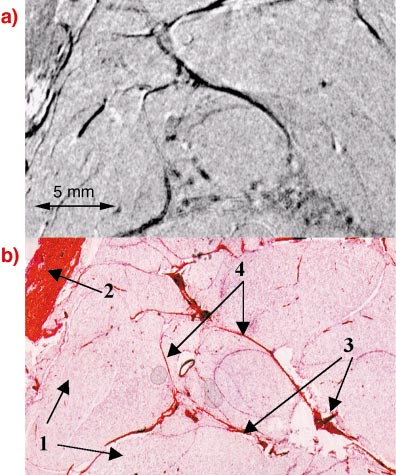
An example for a DEI-CT image of breast tissue invaded by a malignant tumour is shown in Figure 99a and the corresponding stained histological section in Figure 99b. Fine details of the different tissue structures found in the histology can be identified in the DEI image. In particular, the histological examination reveals collagen strands along which cancer cells penetrate into the adipose tissue. Such strands are difficult to visualise in conventional mammography but they are clearly distinguishable in the DEI images.
 |
|
Fig. 99: a) DEI-CT image of cancerous breast tissue showing excellent correspondence with the histological section b) taken at the same position. Arrows 1 indicate adipose tissue, arrow 2 skin and muscular tissue, arrows 3 cancer cells embedded in collagen, and arrows 4 collagen strands. |
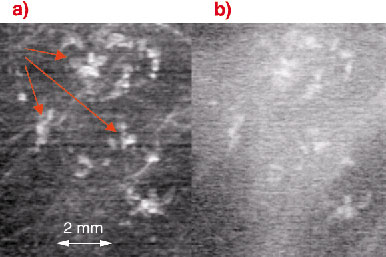
In many cases microcalcifications constitute only benign lesions. Nonetheless, they can be the most important radiological indicator for early breast cancer. In order to distinguish between malignant and benign microcalcifications they have to be classified according to their size, number, morphology, location and distribution. A typical cluster of malignancy indicating microcalcifications, which have variable size and shape and irregular margins, is shown in Figure 100. The analyser-based planar projection image (Figure 100a) allows a much better characterisation than the conventional radiograph (Figure 100b) recorded under the same conditions.
 |
|
|
In connection with this work, standard mammographic phantoms have been imaged with doses lower than 0.1 mGy (mean glandular dose). The potential for low-dose imaging together with the additional diagnostic information obtainable with the DEI method make an extension to human studies envisageable.
References
[1] E.D. Pisano, R.E. Johnston, D. Chapman, J. Geradts, M.V. Iacocca, C.A. Livasy, D.B. Washburn, D.E. Sayers, Z. Zhong, M.Z. Kiss, W.C. Thomlinson, Radiology 214, 895-901 (2000).
[2] F. Arfelli, V. Bonvicini, A. Bravin, G. Cantatore, E. Castelli, L. Dalla Palma, M. Di Michiel, M. Fabrizioli, R. Longo, R.H. Menk, A. Olivo, S. Pani, D. Pontoni, P. Poropat, M. Prest, A. Rashevsky, M .Ratti, L .Rigon, G. Tromba, A. Vacchi, E. Vallazza, F. Zanconati, Radiology 215, 286-293 (2000).
[3] A. Bravin, S. Fiedler, W. Thomlinson; Medical Imaging 2002; Proceedings of SPIE 4682, 167173 (2002).
Authors
S. Fiedler (a), A.Bravin (a), J. Keyrilainen (b), M. Fernandez (a), P. Suortti (c), W. Thomlinson (d), P. Tenhunen (b), P. Virkkunen (b), M-L. Karjalaynen Lindsberg (b), accepted by Physics in Medicine and Biology.
(a) ESRF
(b) Helsinki Central University Hospital (Finland)
(c) University of Helsinki (Finland)
(d) Canadian Light Source (Canada)



