- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2012
- Soft condensed matter
- How YCl3 helps proteins crystallise
How YCl3 helps proteins crystallise
Metastable liquid-liquid phase separation (LLPS) in protein solutions is a fundamental biophysical phenomenon and provides a pathway for biological structure formation [1]. Proteins in solution, like colloids, often interact via effective interactions caused by other components of the solution, i.e. the solvent, salt, etc. Usually the range of the effective attraction for proteins in solution is shorter than their diameter, which leads to a metastable LLPS (corresponding to the gas-liquid phase separation in one-component systems). Close to the LLPS, density fluctuations can significantly reduce the energy barrier of protein crystallisation. In addition to the classical nucleation mechanism, a two-step nucleation mechanism has been proposed [2].
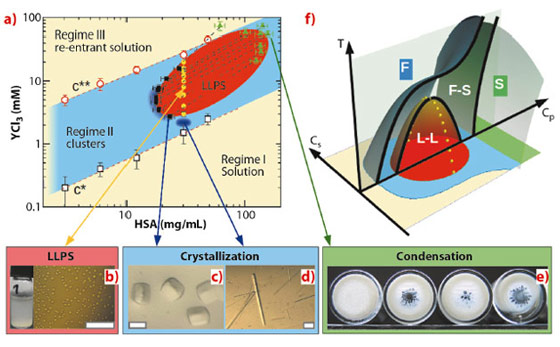
Here we show that trivalent salts can be used to optimise crystallisation conditions for globular proteins and also present some general physical insights. Human serum albumin (HSA) is a medium size globular protein abundant in the blood with a molecular weight of 67 kDa. In Figure 88a, we present the HSA phase diagram as a function of the protein concentration, cP, and the concentration of the trivalent salt YCl3, cs, at 20°C. Trivalent cations tune protein interactions by binding to acidic surface residues, causing a charge-inversion [3,4]. The phase diagram is divided into three regimes. Regime I and III correspond to a transparent single-phase solution and regime II to a turbid two-phase state (Figure 88a). Closer examination of regime II by optical microscopy (Figure 88b) reveals tiny droplets of the protein-rich phase suspended in solution. They merge and grow, indicating a LLPS. A similar LLPS induced by YCl3 is also observed in bovine serum albumin and bβ-lactoglobulin solutions, suggesting LLPS to be a universal phenomenon for negatively charged proteins (46% of the protein family) in the presence of trivalent cations. The partitioning of both protein and salt into two coexisting phases has been determined by X-ray and ultra-violet light absorption. The pairs of coexisting phases connected by tie-lines in Figure 88a define the LLPS coexistence region (in red).
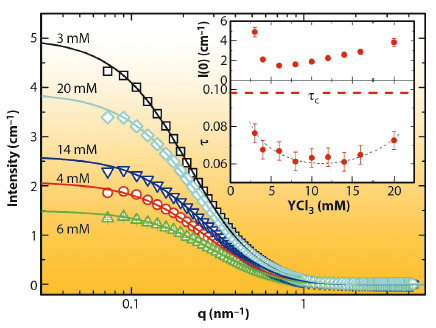
The effective protein-protein interaction upon LLPS has been characterised by SAXS (Figure 89) with a sticky hard sphere structure factor with stickiness τ. The related second virial coefficient B2 indicates a variation in interaction strength throughout the two-phase region as expected for different protein-cation complexes. Approaching the phase boundary, B2 fulfills the thermodynamic criterion for the LLPS critical point as well as lying in the crystallisation regime.
 |
|
Fig. 89: SAXS data with model fitting for the protein-poor phases from sample solutions with initial cp of 31.0 mg mL-1 after LLPS. Only every second data point is plotted for clarity. The insets show τ and the forward intensity I(0) as a function of cs. |
The phase behaviour shown in Figure 88a can now be understood along the usual phase diagram for colloids with short-range attraction. The growth of high quality protein single crystals (Figures 88c and d) supports these theoretical predictions. Importantly, by using multivalent ions, both two-step nucleation (Figure 88c) and classical nucleation close to the LLPS critical point (Figure 88d) can be realised at the same temperature [4,5]. By varying temperature, crystallisation conditions can be further optimised. Several proteins have been previously crystallised using YCl3 as a crucial additive without clarifying its exact role. The presented phase diagram with T, cp and cs as control parameters (Figure 88f) provides a physical understanding of the salt-induced phase behaviour as a pathway towards crystallisation.
Principal publication and authors
F. Zhang (a), R. Roth (b), M. Wolf (a), F. Roosen-Runge (a), M.W.A. Skoda (c), R.M.J. Jacobs (d), M. Sztucki (e) and F. Schreiber (a), Soft Matter 8, 1313-6 (2012).
(a) Universität Tübingen (Germany)
(b) Universität Erlangen-Nürnberg (Germany)
(c) ISIS, Rutherford Appleton Laboratory (UK)
(d) University of Oxford (UK)
(e) ESRF
References
[1] J.D. Gunton, A. Shiryayev and D.L. Pagan, Protein Condensation-kinetic pathways to crystallization and disease, Cambridge University Press, New York (2007).
[2] P. Rein ten Wolde and D. Frenkel, Science 277, 1975-1978 (1997).
[3] F. Zhang, M.W.A. Skoda, R.M.J. Jacobs, S. Zorn, R.A. Martin, C.M. Martin, G.F. Clark, S. Weggler, A. Hildebrandt, O. Kohlbacher and F. Schreiber, Phys. Rev. Lett. 101, 148101 (2008).
[4] F. Zhang, G. Zocher, A. Sauter, T. Stehle and F. Schreiber, J. Appl. Cryst. 44, 755-762 (2011).
[5] F. Zhang, F. Roosen-Runge, A. Sauter, R. Roth, M.W.A. Skoda, R.M.J. Jacobs, M. Sztucki and F. Schreiber, Faraday Discuss. 159, 313 (2012).