- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2012
- Structure of materials
- Trimeron order in magnetite
Trimeron order in magnetite
Magnetite is a naturally-occurring iron oxide mineral with chemical formula Fe3O4. The magnetic properties have been known for more than 2000 years and gave rise to the original concepts of magnets and magnetism. Magnetite has also been the parent for magnetic recording and information storage materials developed since the mid-20th century. In 1939, Verwey [1] discovered that the electrical conductivity of magnetite decreases dramatically at 125 K. It seemed this transition could be understood in terms of the charge states of iron; the localisation of Fe2+Fe3+ states at low temperatures makes the material insulating, and rapid electron hopping between the charge states at higher temperatures gives rise to electronic conductivity. However, despite much effort, the detailed nature of the transition remained controversial because direct structural evidence for localisation of Fe2+ and Fe3+ states in the low temperature state proved elusive [2]. Without a full crystal structure analysis of Fe3O4 below the transition temperature it was not possible to confirm the electronic order.
The low temperature structure of magnetite proved to be remarkably complex. The presence of many small shifts of the atomic positions, of technical problems with crystallographic twinning, and of extinction and multiple scattering effects, have prevented the full structural details from emerging until now. By using a tiny (40 μm) crystal and a focused beam of high energy (74 keV) X-rays at beamline ID11, we were finally able to solve and refine the structure. Cooling the crystal in a magnetic field produced a sample which had a dominant single domain, as shown by the data image in Figure 117. An abundance of diffraction peaks down to a 0.3 Å d-spacing were measured, allowing us to refine a full and accurate structure.
 |
|
Fig. 117: X-ray diffraction intensities from a 40 μm magnetite grain at 90 K, shown for the h = 50 (hkl) plane. The microcrystal is almost single domain; faint spots to the right of the main intensities reveal the secondary twin. |
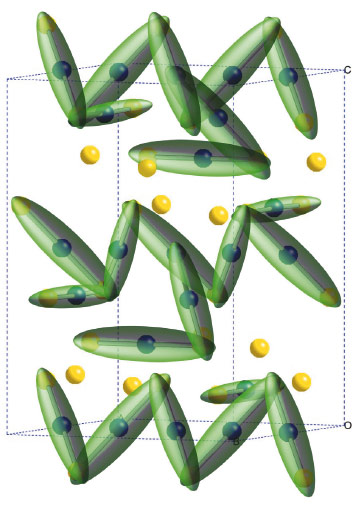
The low temperature distorted structure is very complex, with 24 different iron sites present. Eight of these have tetrahedral FeO4 co-ordination and remain as Fe3+ both above and below the transition. The other 16 sites have octahedral FeO6 co-ordination and have an average valence state of 2.5+ in the high symmetry structure. Our results showed that these split into two groups approximately corresponding to the expected Fe2+ and Fe3+ states. However, we discovered an additional and unexpected interaction. The Fe2+ sites form weak bonds to two of their neighbours so their electron density is spread over all three iron atoms. We describe this unit and the accompanying local distortion as a ‘trimeron’. The low temperature structure of magnetite is thus described by an ordered arrangement of trimerons, as shown on Figure 118. Dynamic motion of trimerons may be important in magnetite above the Verwey transition and to the electronic properties of other transition metal oxides.
 |
|
Fig. 118: Charge order in the low temperature structure of magnetite, with Fe2+/Fe3+ states shown as blue/yellow spheres and the three site trimeron units represented as green ellipsoids. |
Principal publication and authors
M.S. Senn (a), J.P. Wright (b) and J.P. Attfield (a), Nature 481, 173-6 (2012).
(a) Centre for Science at Extreme Conditions and School of Chemistry, University of Edinburgh (UK)
(b) ESRF
References
[1] E.J.W. Verwey, Nature 144, 327–328 (1939).
[2] F. Walz, J. Phys.: Condens. Mat. 14 R285 (2002).



