- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2011
- Structure of materials
- Warming up for clathrates research
Warming up for clathrates research
Scientists have developed a love-hate relationship with clathrates, which have been known for over a century. In oil pipelines, clathrates are seen as a nuisance owing to their ability to block these pipelines. Moreover, ocean floors contain ample quantities of methane clathrates which are thought as a potential future energy source. These materials are also regarded as an alternative route for hydrogen storage for fuel cells. Clathrates were recently in the media when in the Gulf of Mexico oil spill, the recovery efforts were hindered by buildup of methane clathrates. It is clear that these are materials which pose a threat to the energy industry, but also seem promising as a future alternative fuel source. Clathrate hydrates consist of molecular cages in which guest molecules (noble gases and hydrocarbons) can be trapped. Clathrates are usually formed at low temperatures and high pressures.
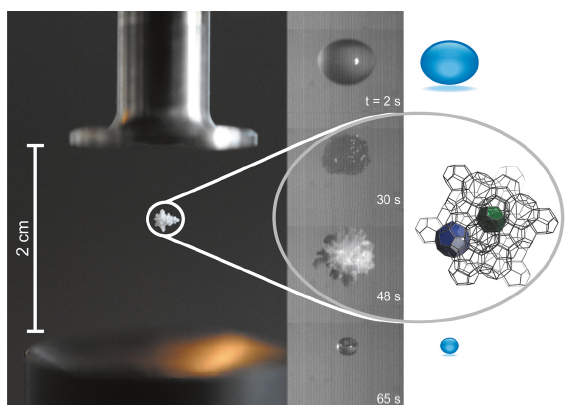
In our experiment, it was possible to form and stabilise clathrate hydrates at ambient conditions for the first time. A small droplet of seed solution (~ 4 µl) was suspended in air by using an acoustic levitator and the formation of clathrate hydrates could subsequently be observed. Due to the efficient evaporation of the seed solution, subzero temperatures were rapidly achieved at the droplet’s surface. A fast camera was used to capture the formation of the clathrate hydrates in situ (see Figure 31). The images show the transition from a transparent droplet to an icy crystalline material and then eventually to a droplet with significantly less volume. It can be seen that the droplet begins to solidify 20-30 seconds after being injected. This process is driven by condensation and subsequent freezing of water on the droplet’s surface. This fast process could be characterised by means of time-resolved X-ray diffraction at beamline ID11. With a time resolution of 500 ms, we obtained diffraction patterns which elucidated the formation process of clathrate hydrate, including the transition from amorphous starting solution to crystalline material.
 |
|
Fig. 31: Clathrate hydrate floating on sound: the series of images shows the different steps during formation of clathrates starting from a suspended droplet. |
The spontaneous formation of clathrates in the acoustic levitator is promoted by a combination of the following ideally suited conditions. The high vapour pressure of the seed solution leads to a low surface temperature. Since the whole surface area of the droplet undergoes fast evaporative cooling, subzero temperatures are reached. Under these conditions atmospheric water condenses on the droplet and an amorphous solid phase is observed. The evaporating seed solution becomes trapped in the amorphous solid that is formed on the surface. The low solubility of the seed solution in water results in rapid supersaturation and clustering of water. Finally, clathrates begin to crystallise and grow. Because of the ambient temperature and pressure conditions, the clathrate only remains stable for a certain period of time and rapidly transforms to water.
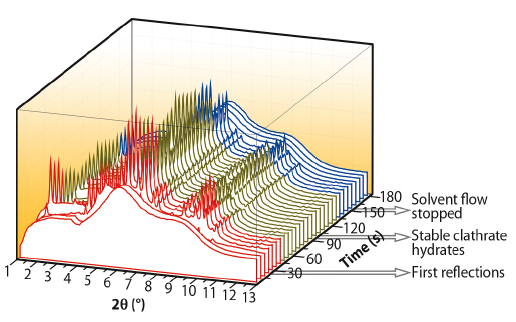
The clathrate hydrates could be stabilised in the suspended droplet by using a piezo-syringe for continuous addition of the seed solution. Hence, using the high time resolution provided by ID11, it was possible to observe the formation process of clathrate hydrates. Figure 32 shows the evolution of the diffraction pattern while adding the seed solution to the suspended droplet. A diffraction pattern of the amorphous phase can be observed initially. After about 30 seconds, the first reflections appear and they remain stable as long as the micro syringe adds solvent to the droplet. Finally, upon stopping the supply of the seed solution, the clathrates are no longer stable and an amorphous diffraction pattern is observed.
 |
|
Fig. 32: Stabilising clathrates at ambient condition: the time-resolved X-ray diffraction patterns show the initial phase of clathrate formation (red area) with appearance of the Bragg peaks, the stabilisation (green) and the dissolution of the clathrates after stopping the supply of seed solution (blue). |
This novel method for the production of stable clathrates at ambient conditions could be employed extensively in clathrate research. As compared to former methods that involved low temperatures and high pressures, the advantages of this experimental approach is the rapid production of clathrates at ambient conditions.
Principal publication and authors
A. Sarfraz (a), M.C. Schlegel (a), J. Wright (b) and F. Emmerling (a), Chem. Comm. 47, 9369-9371 (2011).
(a) BAM Federal Institute for Materials Research and Testing, Berlin (Germany)
(b) ESRF
References
[1] C.E. Taylor and J.T. Kwan, Advances in the study of gas hydrates, Springer (2004).
[2] E.D. Sloan, Nature 426, 353-363 (2003).



