- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2011
- X-ray imaging
- Imaging of antimalarial drugs in Plasmodium falciparum infected red blood cells
Imaging of antimalarial drugs in Plasmodium falciparum infected red blood cells
Few strategies are available for label-free imaging in biological systems with high spatial resolution and high sensitivity. We report for the first time the in vitro localisation of antimalarial drugs in malaria-infected erythrocytes using synchrotron nanochemical imaging. We demonstrate the ability to measure quantitatively the drug distribution inside the parasite that invades red blood cells, particularly in the food vacuole of the parasite. This approach opens new possibilities to improve the understanding of the action mechanisms of antimalarial drugs and resistances.
 |
|
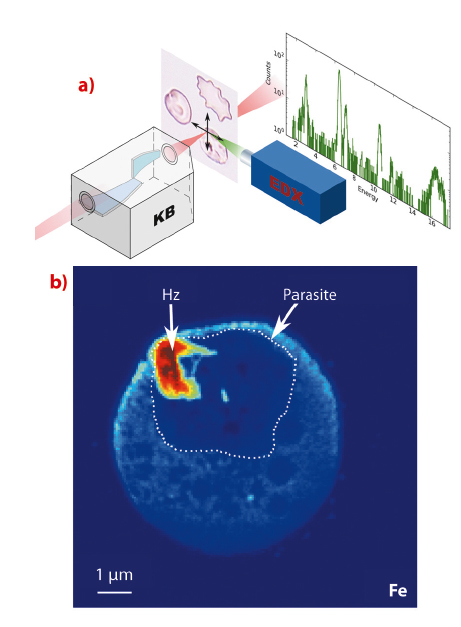
Fig. 126: X-ray fluorescence microscopy of individual red blood cells. a) The sample is scanned through the nanofocus beam produced by the KB optics of the ID22NI station while the X-ray fluorescence signal is being collected. b) Iron fluorescence signal of an infected red blood cell revealing sub-cellular features. The boundary of the parasite is delineated. The malaria pigment (hemozoin Hz) has an increased iron content. |
With approximately 250 million cases and 781,000 deaths reported in 2009, malaria remains the most important human parasitic disease. Among the five Plasmodium species that infect human, P. falciparum is responsible for most cases of severe disease and death, mainly in children under five years of age in Africa. An acute need for new drugs exists as resistance has developed to all antimalarial drugs. Thus, overcoming the problem of drug resistance is an essential goal of antimalarial drug discovery. The new synthetic compound ferroquine (or SSR97193) provides new hope in the fight against malaria. We have used a synchrotron-based nano-imaging technique to localise unlabelled antimalarial drugs inside single red blood cells infected by P. falciparum. The nanoprobe endstation of beamline ID22 allows simultaneous acquisition of the fluorescence signature of most elements of biological interest. Thanks to its exceptionally fine and bright focussed X-ray beam, the end-station permits the imaging of trace elements with a spatial resolution of 50 nm at detection limits down to the attogram level (see Figure 126).
 |
|
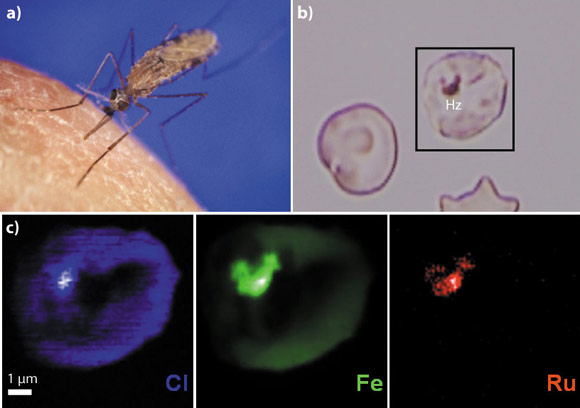
Fig. 127: Nanochemical imaging of an antimalarial drug in an infected red blood cell. a) Mosquito vector of the malaria infectious disease, b) visible light micrograph of the analysed cell showing the brown malaria pigment (hemozoin Hz), c) Sub-cellular distribution of the chlorine (blue), iron (green) and ruthenium (red) atoms. The antimalarial drug ruthenoquine is located in the malaria pigment. |
The parasite digests hemoglobin in the food vacuole, where degraded compounds, potentially toxic to the parasite itself, are crystallised into hemozoin, an insoluble, brown pigment (also called the malaria pigment) that contains different iron species (see Figures 126b and 127b). It is impossible to distinguish the iron atoms of ferroquine, heme, hematin, and hemozoin. Therefore, ruthenoquine, a cold tracer of ferroquine in which the iron atom was substituted by a ruthenium atom, was used as a means to follow the cellular uptake of ferroquine in the infected red blood cells. Ruthenoquine has similar antimalarial activities as ferroquine and accumulates preferentially in infected cells rather than in non-infected cells. We were able to show that ruthenium atoms accumulate in the parasitic food vacuole close to its membranes. Figure 127c shows the distribution of chlorine (blue), iron (green) and ruthenium (red) in an infected red blood cell. It can clearly be seen that the ruthenium atoms are located in the malarial pigment inside the food vacuole. Also, the chlorine atom within antimalarial drugs can be used to image the drug’s uptake without labelling. This was demonstrated not only for ferroquine and ruthenoquine, but also for the long-lived drug chloroquine. Comparing the elemental distributions for the different drugs, the researchers proposed that ferroquine selectively targets the food vacuole through another transport mechanism than the one for chloroquine. This study provides the first demonstration of the localisation of unlabeled antimalarial drugs at pharmalogical doses with high spatial resolution. This strategy improves the understanding of the action mechanisms of already available and novel antimalarial drugs. Moreover, this approach may be applied to a wide range of domains where the quantitative chemical imaging of drugs at the sub-cellular level is critical.
Principal publication and authors
F. Dubar (a), S. Bohic (b,c), C. Slomianny (a), J.C. Morin (d), P. Thomas (d), H. Kalamou (d), Y. Guérardel (a), P. Cloetens (c), J. Khalife (d) and C. Biot (a), Chem. Commun. 48, 910-12 (2012).
(a) Université Lille 1, Villeneuve d’Ascq (France)
(b) Grenoble Institute of Neurosciences, Grenoble (France)
(c) ESRF
(d) Institut Pasteur de Lille, Lille (France)
References
F. Dubar et. al., ACS Chem Biol. 6, 275-87 (2011).



