- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2011
- Dynamics and extreme conditions
- New host for carbon in the deep Earth
New host for carbon in the deep Earth
Carbon exchange occurs between the Earth’s interior and the surface. This deep carbon cycle, taking place over geological timescales of millions of years, has controlled climates of the planet since its formation. However, the extent of recycling and the nature of carbon reservoirs at depths are still poorly known. Carbon is recycled in subduction zones as solid carbonate minerals, their major constituents being magnesite MgCO3 and siderite FeCO3, whereas surficial carbonates are dominated by calcite CaCO3. The pressure-temperature stability of these phases thus plays a key role in the global carbon cycle. Previous studies, focusing on the stability of calcium and magnesium rich carbonates, demonstrated the possibility of carbon recycling into the mantle, e.g. [1]. Thanks to synchrotron radiation studies we have been able to follow the fate of MgCO3 and FeCO3 in the deep Earth.
We performed high pressure – high temperature experiments using a laser-heated diamond anvil cell. Two types of experiments were conducted, with starting materials being either natural carbonate samples (MgCO3 and (Fe,Mg)CO3) or a mixture of oxides (MgO + CO2 and (Fe,Mg)O + CO2). The reacted mixtures and their phase contents were analysed in situ at high-pressure and high-temperature by X-ray diffraction at the high-pressure beamline ID27. Ex situ analyses by scanning transmission electron microscopy - electron energy loss spectroscopy (STEM-EELS) were performed on the samples recovered at ambient conditions.
 |
|
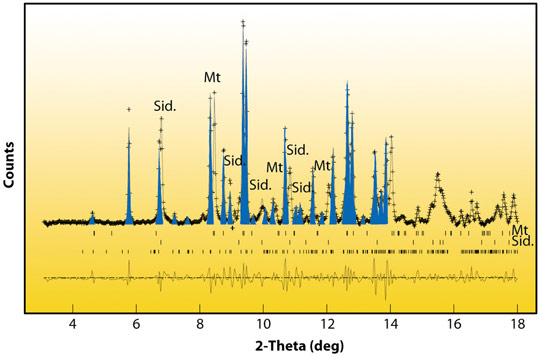
Fig. 20: X-ray diffraction pattern of (Fe,Mg)CO3 collected at 80 GPa and ~2050 K. Blue peaks highlight diffraction peaks of the new high-pressure phase. Sid. refers to the siderite-rich starting material (Fe,Mg)CO3, Mt to magnetite (Fe3O4). |
New X-ray diffraction peaks (in blue in Figure 20) were observed for all compositions, indicating the existence of a new high-pressure phase of MgCO3 and (Fe,Mg)CO3 at pressures exceeding 80 GPa and temperatures above 1850 K. These conditions correspond to depths larger than 1,800 km in the Earth’s mantle. A new silicate-like crystal structure has been discovered (see Figure 21). It is based on (CO4)4- groups instead of (CO3)2- molecules characteristic of low pressure carbonates. This phase is close to that predicted by first principle calculations [2]. Ex situ STEM-EELS analyses coupled to Density Functional Theory (DFT) calculations confirmed that the peculiar spectroscopic signature at the carbon K-edge is explained by the presence of (CO4)4- groups. The implications of this new phase are far reaching because its solubility in silicates and its melting behaviour are likely to be different from those of low-pressure carbonates. These properties remain yet to be determined.
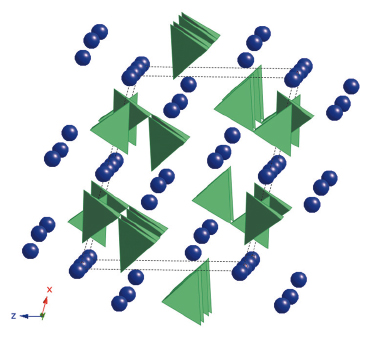 |
| Fig. 21: Structure of the new high-pressure phase. (CO4)4- tetrahedra appear in green and magnesium or ferric iron atoms are shown as violet spheres. |
Furthermore, iron revealed an interesting behaviour in these experiments: the normal state of iron in carbonates, ferrous iron (labeled Fe2+ or Fe(II)), was found to be unstable in the presence of carbonate ions. Fe(II) was oxidised into ferric iron (Fe3+ or Fe(III)), coupled to reduction of carbon from CO2 or carbonate ions into carbon monoxide (CO) and nano-diamonds, as shown by EELS performed at the C-K edge on samples recovered at ambient conditions. Those oxidation-reduction reactions explain the presence of magnetite Fe3O4 (Mt in Figure 20) in the high-pressure X-ray diffraction patterns. Moreover, ex situ STEM-EELS analyses showed that iron in the new high-pressure phase (Figure 21) was indeed Fe(III). These reactions provide a possible new mechanism for driving carbon-bearing minerals down to the core-mantle boundary of the Earth and for generating ultra-deep diamonds within an oxidised subducted carbonate starting material.
A new picture of the evolution of carbon in the deep Earth is thus emerging. If subducted iron and magnesium carbonates are sufficiently isolated from the surrounding mantle, for instance, in a relatively cold subducted slab, they might be transported beyond 1,800-km depth. There, they will transform into the newly observed phase and diamonds will be produced as well. The validity of the proposed mechanism needs to be further studied in actual silicate-bearing mantle assemblages and within minute inclusions found in ultra-deep diamonds coming from the lower mantle.
Principal publication and authors
E. Boulard (a,b), A. Gloter (c), A. Corgne (a,d), D. Antonangeli (a), AL. Auzende (a), J.P. Perrillat (e,f), F. Guyot (a) and G. Fiquet (a), Proceed. Nat. Acad. Sciences, 108, 5184-5187 (2011).
(a) IMPMC, IPGP, Université Pierre et Marie Curie, CNRS, Université Paris Diderot (France)
(b) Extreme Environments Laboratory, Stanford University (USA)
(c) Laboratoire de Physique des Solides, Université Paris Sud 11, CNRS (France)
(d) Institut de Recherche en Astrophysique et Planétologie, Université de Toulouse, CNRS (France)
(e) ESRF
(f) Géologie de Lyon, Université Claude Bernard, ENS Lyon, CNRS (France)
References
[1] S. Poli, E. Franzolin, P. Fumagalli and A. Crottini, Earth Planet. Sci. Lett., 278, 350-360 (2009).
[2] A.R. Oganov, S. Ono, Y.M. Ma, C.W. Glass and A. Garcia, Earth Planet. Sci. Lett., 273. 38-47 (2008).



