- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2011
- Dynamics and extreme conditions
- Role of platinum in tin oxide based gas sensors: a study under working conditions
Role of platinum in tin oxide based gas sensors: a study under working conditions
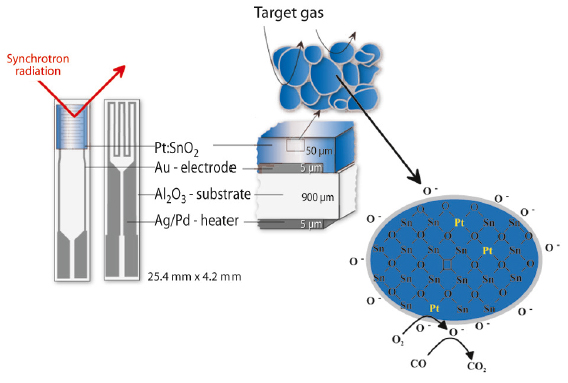
Chemical gas sensors based on semiconducting metal-oxides are currently one of the most abundant and best studied types of sensor device for the detection of toxic gases and pollutants. The state of the art design of these sensors comprises the deposition of the polycrystalline porous metal-oxide film onto a ceramic substrate (see Figure 16). To monitor the electrical changes as a function of the surrounding atmosphere, these substrates are equipped with electrical contacts and a heater. In most cases, minute quantities of noble metals (Pt, Pd and Au) are added in the first synthesis steps to the metal-oxide in order to tune their selectivity, lower the operation temperature and improve the response time.
 |
|
Fig. 16: Schematic view of the sensor device. The cartoon demonstrates the role of the Pt additives in sensing carbon monoxide with SnO2 based gas sensors. |
Although this “doping” technique has been known since the early days of metal-oxide based gas sensors, there is still a strong discussion about how these additives contribute to the improved performance. The chemical state of the additives, their distribution in the material and whether they change structurally during the sensing procedure is still a matter of debate [1]. This had not been studied under real sensing conditions because experimental requirements are very challenging: the platinum from the electrodes and the heater of the device would dominate the information from the platinum additive in the sensing layer. In the present study, carried out at beamline ID26, this obstacle has been overcome by a new design of the substrate. Gold was employed instead of platinum to produce the ohmic contacts, and an alloy of Pd and Ag was used instead of Pt for the heater. To eliminate the disturbing fluorescence lines of gold and to detect the small amount of 0.2 wt.% Pt in the 50 µm thick SnO2 sensing layer, the generally-used solid-state detector was replaced by an X-ray emission spectrometer [2]. This allowed the effect of the lifetime broadening at the Pt L3-edge to be minimised and thus high energy-resolution fluorescence-detected X-ray absorption spectra on the modified sensor devices were successfully recorded. The state of Pt and its role in sensing could consequently be investigated under real sensor operation conditions (exposure to reducing gases at a temperature of 300°C).
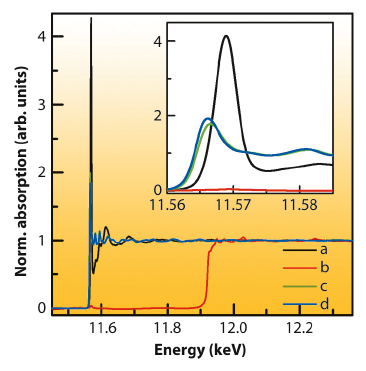
Figure 17 presents the Pt L3-XAS spectra. Comparing the spectra of the Pt doped SnO2 with those of the metallic Pt and PtO2 references, one can clearly conclude, that Pt in the sensing layer is in an oxidised state (almost identical to the spectra of the PtO2). The stronger white line intensity when compared to the spectra of PtO2 illustrates that Pt is even in a more ionic state than in the reference. Furthermore, the evaluation of the range-extended (beyond the Au edge) EXAFS data indicates that Pt is at an atomic level incorporated in the matrix of the metal-oxide. It seems that some of the Sn sites are substituted by Pt during the synthesis.
 |
|
Fig. 17: Pt L3 edge EXAFS spectra of a 0.2 wt % Pt:SnO2 sensor with Au electrodes measured in (a) high energy resolved fluorescence detection (HERFD) and (b) traditional detection mode where the spectrum is dominated by the Au edge at 11.9 keV; as reference HERFD-XANES spectra of a sensor with Pt electrodes (c) and Pt foil (d) are shown. |
The role and state of the Pt additives was further investigated by recording XAS spectra simultaneously with resistance changes during target gas exposure (H2 and CO) at different operation temperatures (200-400°C). Only minor changes in the electronic structure could be observed, while large resistance changes were detected upon CO and H2 exposure in air.
The improved sensing properties of Pt doped SnO2 sensors were previously attributed to either metallic Pt particles or clusters on the surface of the metal-oxide. This assumption can now be excluded because we were able to demonstrate that Pt is in its oxidised state even under harsh reducing conditions. The studies further indicate that Pt substitutes some of the Sn sites in the SnO2 matrix. The overall improved selectivity and sensitivity of the Pt doped SnO2 can therefore be explained by a combination of a surface effect, namely the creation of new adsorption sites for oxygen (see Figure 16), and by an additional bulk effect. The role of Pt in metal oxide base gas sensors is therefore more complex than initially anticipated.
Principal publication and authors
M. Hübner (a), D. Koziej (b), M. Bauer (c), N. Barsan (a), K. Kvashnina (d), M.D. Rossell (b), U. Weimar (a) and J.-D. Grunwaldt (c), Angew. Chem. Int. Ed. 50, 2841–2844 (2011).
(a) Faculty of Science, Institute of Physical Chemistry, Tuebingen University (Germany)
(b) Department of Materials, ETH Zürich (Switzerland)
(c) Institute for Chemical Technology and Polymer Chemistry, Karlsruhe Institute of Technology (Germany)
(d) ESRF
References
[1] D. Koziej, M. Hübner, N. Barsan, U. Weimar, M. Sikora and J.D. Grunwaldt, Phys. Chem. Chem. Phys. 11-38, 8620-8625 (2009).
[2] P. Glatzel, F.M.F. de Groot and U. Bergmann, SRN 22, 12-14 (2009).



