- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2011
- Dynamics and extreme conditions
- Cerium’s unusual behaviour
Cerium’s unusual behaviour
When a solid is subjected to high pressure, its atomic arrangement may simply contract continuously or it might change abruptly at some point. For most elements, the transition is accompanied by an abrupt decrease in volume, which usually would imply that a single crystal crushes into a poly-crystal or a powder. However, recent high-pressure and high-temperature X-ray diffraction experiments on pure cerium –the most abundant rare-earth element, with applications ranging from catalysts to fluorescent lamps– reveal that this element does not conform to this picture.
Diffraction data refinements do not provide any evidence in favour of a possible structure distortion of cerium along the γ![]() α transformation, irrespective of the poly- or single-crystalline nature of the samples. In a unique manner among all the elements of the periodic table, the phase transition in cerium can be described as an isomorphic first-order transition with a volume collapse of about 15% at 300 K. Moreover, the non-equilibrium phase diagram of cerium confirms the entropic term T∆S to be the predominant driving force of the transition, in agreement with the notion that the γ
α transformation, irrespective of the poly- or single-crystalline nature of the samples. In a unique manner among all the elements of the periodic table, the phase transition in cerium can be described as an isomorphic first-order transition with a volume collapse of about 15% at 300 K. Moreover, the non-equilibrium phase diagram of cerium confirms the entropic term T∆S to be the predominant driving force of the transition, in agreement with the notion that the γ![]() α transition is controlled by the behaviour of the single 4f electron, archetypical paradigm of the localisation-delocalisation phenomenon encountered in f-electron systems.
α transition is controlled by the behaviour of the single 4f electron, archetypical paradigm of the localisation-delocalisation phenomenon encountered in f-electron systems.
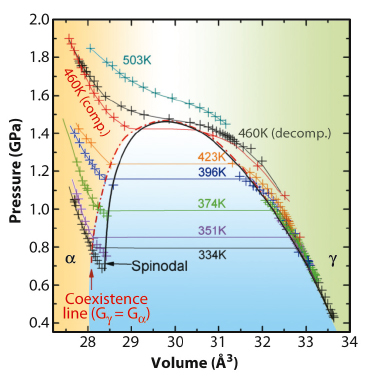
Surprisingly, this X-ray diffraction study has confirmed that the γ![]() α transition line ends at a solid-solid critical point (460 K and 1.44 GPa) where the pressure dependence of the lattice parameter shows a vertical tangent. The analogy between the experimental p(V) curves given in Figure 10 and the liquid-gas transition is obviously tempting. The γ
α transition line ends at a solid-solid critical point (460 K and 1.44 GPa) where the pressure dependence of the lattice parameter shows a vertical tangent. The analogy between the experimental p(V) curves given in Figure 10 and the liquid-gas transition is obviously tempting. The γ![]() α transformation in cerium can indeed be pictured in the framework of a generic description where the most probable atomic configuration of the f electron in γ-cerium is a single occupied state, corresponding to a gas-like regime. Conversely, the α-phase can be pictured as a “liquid” where the probability of double occupancy is calculated to be significant.
α transformation in cerium can indeed be pictured in the framework of a generic description where the most probable atomic configuration of the f electron in γ-cerium is a single occupied state, corresponding to a gas-like regime. Conversely, the α-phase can be pictured as a “liquid” where the probability of double occupancy is calculated to be significant.
 |
|
Fig. 10: Clapeyron diagram of cerium along isotherms. The coexistence (dashed-dotted red) line delimits the region where the system is in a biphasic configuration, whereas the spinodal line (dashed–dotted black line) represents the boundary between metastable and unstable conditions. |
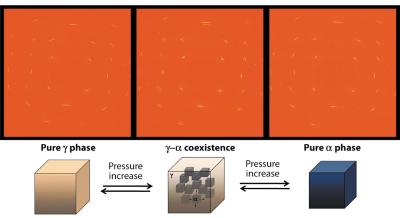
The angular dispersive X-ray diffraction patterns collected in runs using single-crystalline samples allowed us also to address the transformation mechanisms of the γ![]() α transition in detail. Here again, cerium behaves in very counter-intuitive ways when subjected to high pressures: at temperatures around ambient conditions, both the single crystalline nature and the orientation of the crystals are preserved in spite of the 15% volume collapse across the phase transition. Even more intriguing, the occurrence of two single-crystals in coexistence with the same structure and orientation (Figure 11) is recorded in the metastable region. All these features clearly call for a fascinating scenario of a diffusionless phase transformation induced by the strain energy. This picture is consistent with the very large softening of the longitudinal elastic modulus and the absence of effects on the shear modulus [1], but contrasts to what is usually observed in a purely martensitic transformation. Though such phenomenological description has never been previously proposed for any kind of element, we emphasise that a dislocation-based mechanism could sustain this conjecture. Since the α and γ crystals remain in crystallographic registry, there must be a large number of edge dislocations, possibly forming the periodic array of misfit necessary to accommodate the large volume difference [2].
α transition in detail. Here again, cerium behaves in very counter-intuitive ways when subjected to high pressures: at temperatures around ambient conditions, both the single crystalline nature and the orientation of the crystals are preserved in spite of the 15% volume collapse across the phase transition. Even more intriguing, the occurrence of two single-crystals in coexistence with the same structure and orientation (Figure 11) is recorded in the metastable region. All these features clearly call for a fascinating scenario of a diffusionless phase transformation induced by the strain energy. This picture is consistent with the very large softening of the longitudinal elastic modulus and the absence of effects on the shear modulus [1], but contrasts to what is usually observed in a purely martensitic transformation. Though such phenomenological description has never been previously proposed for any kind of element, we emphasise that a dislocation-based mechanism could sustain this conjecture. Since the α and γ crystals remain in crystallographic registry, there must be a large number of edge dislocations, possibly forming the periodic array of misfit necessary to accommodate the large volume difference [2].
In conclusion, these sets of experiments, carried out at beamline ID09, clearly show that the surprising phase transition and transformation mechanism in cerium can be understood on the basis of crystallographic and thermodynamic arguments. These observations represent an important step forward in achieving a reliable and unambiguous picture of the mechanism of phase transformation in cerium, an element archetypical of the localisation-delocalisation phenomenon encountered in f-electron systems, including plutonium and other actinides.
Principal publication and authors
F. Decremps (a), L. Belhadi (a), D.L. Farber (b), K.T. Moore (b), F. Occelli (c), M. Gauthier (a), A. Polian (a), D. Antonangeli (a), C.M. Aracne-Ruddle (b), and B. Amadon (c), Phys. Rev. Lett. 106, 65701 (2011)
(a) IMPMC, Université Pierre et Marie Curie, Paris (France)
(b) LLNL, Livermore, California (USA)
(c) CEA, DAM, DIF, Arpajon (France)
References
[1] F. Decremps, D. Antonangeli, B. Amadon and G. Schmerber, Phys. Rev. B 80, 132103- (2009).
[2] K.T. Moore, L. Belhadi, F. Decremps, D.L. Farber, J.A. Bradley, F. Occelli, M. Gauthier, A. Polian and C.M. Aracne-Ruddle, Acta Materialia 59, 6007-6016 (2011).