- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Structural biology
- Discovery of a marine bacterial porphyranase: from gene to structure and beyond
Discovery of a marine bacterial porphyranase: from gene to structure and beyond
The recent availability of genomic data on heterotrophic marine bacteria emphasises the crucial role that microbes play in the global carbon cycle. However, the massive presence in these genomes of proteins of unknown function hampers our understanding of the detailed mechanisms by which this carbon cycle is fine-tuned. Moreover, genome data from marine organisms are essentially annotated in light of the biochemical knowledge accumulated on bacteria and fungi decomposing terrestrial plants. But marine algal polysaccharides clearly differ from their terrestrial counterparts [1] and their associated enzymes usually constitute novel protein families, as described in the carbohydrate-active enzyme database (CAZy, http://www.cazy.org).
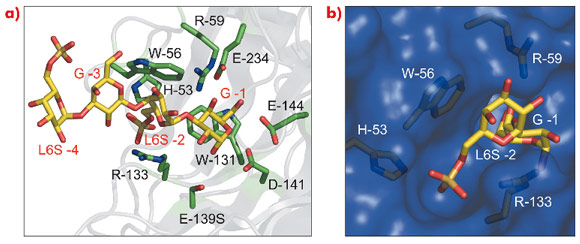
We have applied a knowledge-based strategy for the structural and biochemical characterisation of candidate ‘hypothetical conserved proteins’ or members of phylogenetic sub-families to identify new enzyme functions, missing in the metabolic pathways by which marine polysaccharides are degraded in the marine environment. In this way we selected several potential enzymes within the GH16 polyspecific glycoside hydrolase family with the aim of expressing them in E. coli and of performing enzymatic activity screening. Two of the enzymes selected displayed high activity, measured through the release of reducing sugars from a specific red algal polysaccharide called porphyran. Their crystal structures, in complex with substrate molecules, were then determined using diffraction data collected at the ESRF (ID23, ID14-1 and BM30A). These allowed the unambiguous assignment of residues responsible for the new substrate specificity (Figure 104), the hydrolytic cleavage of porphyran.
Curious about the distribution of this enzymatic activity in the environment, we screened GenBank using BLAST searches to find homologous proteins having the crucial residues that make them porphyranases. To our surprise, these enzymes were not only present in marine bacteria but also in B. plebeius, a human gut bacterium, isolated from Japanese individuals. In depth phylogenetic analyses and pushing our BLAST screening to include recently available metagenomic data collected from soil, ‘open ocean’ or human gut of North American [2] and Japanese [3] individuals revealed that the presence of these enzymes was currently restricted to coastal marine bacteria – and to a single human gut species!
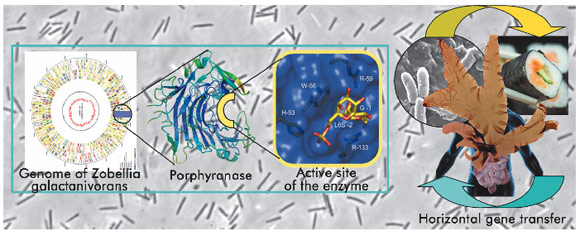
Where was the link? We know from many other studies that intestinal microorganisms, collectively known as the intestinal ‘microbiota’ are, to a large extent, specialised in digesting dietary fibres, such as starch and cellulose, for which the human genome does not contain any degradative enzymes. It is likely that significant competition occurs for these substrates. Indeed, genes dedicated to carbohydrate acquisition and degradation, such as those encoding glycoside hydrolase enzymes, are prominent in metagenomic data sets of gut microbiota and are also enriched in the genomes of many species residing in the intestine, including members of the phylum Bacteroidetes [4]. Our work shows that one apparent strategy for success of bacteria in the intestinal ecosystem is to become proficient at using the dietary polysaccharides that the host consumes. This proficiency can be acquired through lateral gene transfer, a strategy known to be at the basis of evolutionary adaptation. In our example, the detailed analyses of the gene context of the porphyranases, present only in marine and gut bacteria, shows that a horizontal gene transfer – from a marine bacterium to a human gut bacterium has most probably occurred. We believe that seaweed, which is prevalent in the Japanese diet – including the Porphyra-derived nori, used to wrap sushi – was probably the source of the marine microorganism that introduced the useful genes acquired by the human gut bacterium (Figure 105).
 |
|
Fig. 105: Schematic representation of our strategy ‘from the genome to the structure’ and the horizontal gene transfer between marine and Japanese human gut bacteria via the red algae Porphyra. |
Principal publication and authors
J.H. Hehemann, G. Correc, T. Barbeyron, W. Helbert, M. Czjzek and G. Michel, Nature 464, 908-912 (2010).
Végétaux Marins et Biomolécules, UMR7139 CNRS-UPMC, Station Biologique de Roscoff (France)
References
[1] B. Kloareg and R.S. Quatrano, Oceanogr Mar Biol Ann Rev 26, 259-315 (1988).
[2] P.J. Turnbaugh, M. Hamady, T. Yatsunenko et al. and J.I. Gordon, Nature 457, 480-4 (2009).
[3] K. Kurokawa, T. Itoh, T. Kuwahara et al. and M. Hattori, DNA Research 14, 169-81 (2007).
[4] E.C. Martens, N.M. Koropatkin, T.J. Smith and J.I. Gordon, J. Biol. Chem. 284, 24673-24677 (2009).