- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Structure of materials
- Fullerenium salts: a new family of C60 based materials
Fullerenium salts: a new family of C60 based materials
Fullerides are C60 charge transfer salts that have notable electronic transport and magnetic properties, like high-Tc superconductivity [1], molecular magnetism, and ionic conductivity [2]. These compounds, usually intercalated with alkali or alkaline-earth ions, always contain C60 in its reduced (negative, anionic) state. Analogous compounds where fullerene is oxidised -in the positive, cationic state- referred to as fullerenium salts, are difficult to synthesise owing to the high fullerene oxidation potentials and to the marked electrophilic propensity of C60n+. Nevertheless, theoretical calculations suggest that in such systems a huge increase of the superconducting critical temperature should be expected with respect to that observed in anionic fullerides.
Here we describe the first preparation and the structural characterisation of a member of this new class of C60 based materials, whose formula is C60(AsF6)2. Its synthesis was achieved thanks to the oxidising power of the strong Lewis acid AsF5 and also the weak nucleophilic character of its conjugate base that allowed the stabilisation of the highly reactive C602+ cations, which unexpectedly organise themselves to form a novel 1D polymer in the crystalline state.
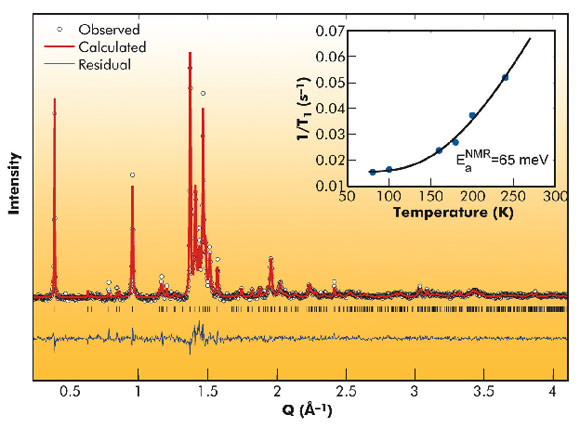
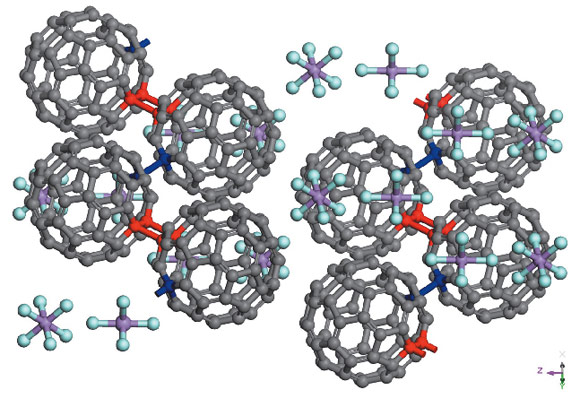
High-resolution synchrotron and neutron powder diffraction were necessary to achieve a definitive structural identification of this material. At room temperature, both techniques showed the presence of a single phase, which was indexed with an orthorhombic cell (a = 10.450(3) Å, b = 9.993(6) Å, c = 32.07(1) Å), although the study of the molecular configuration has proven that the true symmetry is monoclinic, space group I2/m with β ≈ 90°. The density of the compound, determined by gas picnometry, of 2.2(2) g cm-3 suggested that the correct stoichiometry is C60(AsF6)2. Rietveld refinement was performed at the same time on both synchrotron (see Figure 48) and neutron data: while the former allowed localisation of the various components of the cell, the latter permitted the determination of the precise molecular orientation. After trials with different dispositions compatible with the symmetry group, the agreement factors invariably improve after ordering the C60 units in an unusual zigzag configuration, as shown in Figure 49. This arrangement is compatible with the presence of both alternated four-membered carbon rings ([2+2] cycloaddition) and single C-C bonds between the fullerene molecules along the zigzag chain.
Nuclear magnetic resonance (see inset of Figure 48), SQUID magnetometry and DC conductivity show that this polymer is a diamagnetic semiconductor while the lack of the theoretically predicted high Tc superconductivity could be ascribed to the strong reduction of crystal and molecular symmetry upon polymerisation. Nonetheless, the small value of the energy gap (approx. 65 meV) could suggest the proximity of a metallic phase.
The higher density of states (DOS) at the Fermi energy of the 5-fold-degenerate h1u HOMO derived band, as compared to the 3-fold-degenerate LUMO involved in the anionic fullerides, leads to the prediction of a substantial increase of the critical temperature in a hypothetical cubic superconducting fullerenium compound. However, our evidence suggests that the high reactivity of C602+ ions triggers the formation of intermolecular bonds. One can hypothesise that these compounds would have been polymeric in their hole-doped state and consequently hardly superconducting.
In conclusion, the successful synthesis of the first fullerenium salt in the solid state highlights the marked propensity of C60 cations to polymerise. The polymerisation also suppresses the expected exotic properties of a hole-doped C60 compound. The preparation of a monomeric compound either by thermal depolymerisation of the present system or by co-intercalation of inert molecules is now under study.
Principal publication and authors
M. Riccò (a), D. Pontiroli (a), M. Mazzani (a), F. Gianferrari (a), M. Pagliari (a), A. Goffredi (a), M. Brunelli (b), G. Zandomeneghi (c), B. H. Meier (c) and T. Shiroka (d), J. Am. Chem. Soc. 132, 2064 (2010).
(a) Dipartimento di Fisica, Università di Parma (Italy)
(b) Institut Laue Langevin, Grenoble (France)
(c) Physical Chemistry Laboratory, ETH-Zurich (Switzerland)
(d) Laboratorium für Festkörperphysik, ETH-Zurich (Switzerland)
References
[1] Y. Ihara, H. Alloul, P. Wzietek, D. Pontiroli, M. Mazzani and M. Riccò, Physical Review Letters 104, 256402 (2010).
[2] M. Riccò, M. Belli, M. Mazzani, D. Pontiroli, D. Quintavalle, A. Janossy and G. Csanyi, Physical Review Letters 102, 145901 (2009).