- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Structure of materials
- In situ diffraction of the electrochemical reduction pathway of TiO2
In situ diffraction of the electrochemical reduction pathway of TiO2
It is widely thought that an electrochemical method will succeed the costly Kroll process, which is used to make the majority of titanium in the world. In 2000, Professors Fray, Farthing and Chen discovered a method of electrochemically deoxidising metal oxide in a CaCl2 melt, in a process now known as the FFC Cambridge Process [1]. Since 2000, there has been significant research into understanding the reduction process for elements such as Ti, Ta, Zr, Cr, etc. The FFC Cambridge process also has the potential to form alloys, such as Ti-6Al-4V, by co-reducing metal oxides, such as TiO2, Al2O3 and V2O5.
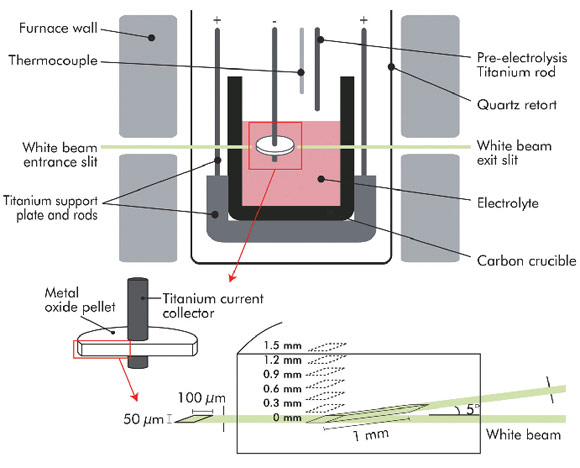
This discovery has encouraged a broader exploration of electrochemical reduction as a competitive extraction route, which has the potential to replace current commercial extraction routes for elements like Ti, Ta, Zr, etc. Whilst reducing these metal oxides to metal is relatively simple, doing it consistently to form a product with low concentrations of impurities in an efficient way has proven to be more challenging. To achieve these goals, it is important to understand fundamentally the reactions taking place, so that it becomes possible to control the process. Unfortunately high temperature electrochemical studies (as shown in Figure 44), do not lend themselves to conventional analysis techniques.
 |
|
Fig. 44: Electrochemical reduction cell showing the sampling volumes employed. |
Titanium is one of more difficult metals to produce from the FFC Cambridge process owing to the various phases that form during reduction; however, it remains one of the most lucrative. As such, titanium oxide reduction to titanium has been the most studied system with regards to the FFC Cambridge process. However, to date, studies were based on a combination of ex situ phase studies and voltammetric techniques, which have resulted in an ambiguous understanding of the reduction behaviour and role of intermediary phases. For instance, the formation mechanism for the intermediate phases CaTiO3 and CaTi2O4 and TiC was much debated, with no clear evidence to show whether it was chemically or electrochemically formed.
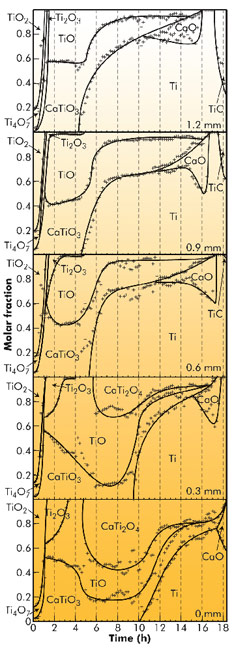
In 2008, the authors proposed an experiment at ID15A where the phase evolution during the FFC Cambridge reduction of titanium dioxide to titanium metal could be logged in situ. The experiment would require a redesign of the classic FFC cell to make it X-ray transparent whilst being capable of completing an eighteen hour FFC Cambridge reduction. The diffraction patterns were analysed using Rietveld refinement and volume fraction and phase information was extracted, resulting in the phase map shown in Figure 45.
 |
|
Fig. 45: Rietveld-refined phase evolution throughout the pellet. Centre -0 mm to near-surface, 1.2 mm. |
The most significant results are as follows. A reduction pathway from TiO2 to Ti was presented and the formation of the phases CaTiO3, CaTi2O4 and TiC were discussed. CaTiO3 was found to result from incorporation of CaO from the molten salt medium into titanium Magneli phases. CaTi2O4 was found to form as a comproportionation reaction between TiO and CaTiO3, though this was found to occur only in the presence of Ti. It was found that TiC was formed via an electrochemically assisted route, i.e. carbon was electrochemically deposited onto the Ti which chemically reacted to form TiC.
The deoxidation of Ti could be interpreted by combining thermal coefficient of expansion values with data from the literature on the effect of interstitial O on the lattice parameter of Ti. Subsequently it was possible to correlate the formation of TiC to a critical value of interstitial O in Ti (14 at.% O). This discovery has the potential to enable commercial production of ASTM grade Ti powders.
This study demonstrates that high-energy synchrotron radiation can be used as a powerful tool to analyse electrochemical processes such as the FFC Cambridge process. Owing to the high temperature of operation (900°C) and the experimental conditions (argon atmosphere and corrosive molten salt) of the FFC process, analysis of the process thus far has been conducted through a combination of ex situ phase studies and voltammetric studies. Unfortunately this had competing interpretations of results and a number of competing hypotheses for the evolution of phases and their role during reduction. This study presents an unequivocal understanding of the FFC Cambridge process by analysing the in situ phase evolution. This study has furthered the understanding of the process and has accelerated the prospect of commercially available FFC produced Ti powders.
Principal publication and authors
R. Bhagat (a), D. Dye (b), S.L. Raghunathan (b), R.J. Talling (b), D. Inman (b), B.K. Jackson (b), K.K. Rao (c) and R.J. Dashwood (a), Acta Materialia 58, 5057–5062 (2010).
(a) University of Warwick, Coventry (UK)
(b) Imperial College, London (UK)
(c) Metalysis, Rotherham (UK)
References
[1] G. Chen, D. Fray and T. Farthing, Nature 407, 361-4 (2000).



