- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Electronic structure and magnetism
- EXAFS and XRD characterisation of palladium sorbents for high temperature mercury capture from fuel gas
EXAFS and XRD characterisation of palladium sorbents for high temperature mercury capture from fuel gas
The gasification of coal is a potentially important way of utilising coal reserves, for example in integrated gasification combined cycle (IGCC) power plants or in the production of syn-gas for chemicals. The syn-gas or fuel gas (CO, CO2, H2) derived from this process contains low concentrations of heavy metals that were present in the coal and become volatile during gasification. Release of mercury vapour in its elemental form can lead to environmental and technical problems during fuel/syn gas utilisation.
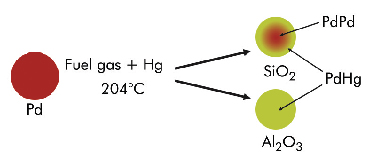
Palladium can be used for mercury vapour removal from fuel gas at elevated temperatures [1]. The overall thermal efficiency of an IGCC system is improved with removal of mercury at higher temperatures using sorbents capable of operating at these temperatures. The sorbent samples Pd/Al2O3 and Pd/SiO2 have significantly different Hg adsorption capacities at operating temperatures, with higher Hg capacities for the Pd/SiO2 sorbent than Pd/Al2O3, although Pd dispersions are higher for the Pd/Al2O3 sorbent. A detailed structural investigation using complimentary chemical and physical (XRD, EXAFS) methods was undertaken to understand whether the mercury is adsorbed on the support or interacts with palladium leading to the formation of an alloy-type material.
Long range order as determined by ex situ XRD shows significant differences in the nature of the alloys formed on the two sorbents following exposure to Hg in a syn gas feed at 204°C. The Pd/Al2O3 sorbent produced a single homogeneous solid solution of Pd-Hg consistent with the limited literature reports on the Hg-Pd phase diagram, whilst the silica supported Pd produced an alloy of varying composition.
Pd K and Hg L3-edge EXAFS data were collected at 20 K on the BM29 beamline. Consistent with ex situ XRD, the EXAFS analysis (both Pd and Hg edge) of the Pd/Al2O3 sorbent exposed to fuel gas containing mercury can be interpreted as a Pd–Hg alloy phase. The near neighbour distances obtained from both the Pd and Hg edge EXAFS were found to be very similar suggesting a uniform alloy was formed. However, the most striking difference between the Pd K-edge and Hg L3-edge results was that Pd is surrounded predominantly by Pd whilst the Hg is surrounded by Pd; no significant Hg–Hg contacts (from Hg L3-edge EXAFS) or Pd–Hg contacts (from Pd K-edge EXAFS) were observed. This is most likely due to the low concentrations of mercury in the system and, in addition, the mercury atoms being randomly distributed throughout the palladium lattice in such a way that palladium atoms are mainly in contact with palladium (Figure 85).
 |
|
Fig. 85: Proposed Pd-Hg interactions as determined by XAS and XRD from silica and alumina sorbents. |
In contrast, Hg exposed Pd/SiO2 sorbent showed markedly different results. The refined Hg–Pd distance from the Hg L3-edge, supported by ex situ XRD indicates that there is alloy formation similar to that in the Hg exposed Pd/Al2O3 sample. However, the Pd K-edge data suggest there are mainly Pd–Pd scatterers present. In particular the first neighbour distances obtained from Pd K-edge data differ by ca. 0.03 Å compared to Hg L3-edge results and the Pd–Pd distance was very close to that obtained for untreated Pd/SiO2 material. On the other hand the Hg L3-edge results are similar to those obtained for the Hg exposed Pd/Al2O3 system indicating that there is a Hg–Pd alloy present in the system. This suggests a more complex structure to the Pd–Hg particles than is present in the Hg exposed Pd/Al2O3 sample. One model consistent with the XRD and EXAFS data is a core in the metal particles that contains predominantly Pd–Pd neighbours with the periphery of the particle consisting of Pd–Hg neighbours. It is likely, therefore, that there is a gradual change in composition with Pd–Hg alloy in the near surface part of the metal particles, the Hg concentration progressively decreasing through the particle leading to a Pd metal particle-like phase dominating the core.
In conclusion, detailed XRD and XAS studies reveal that, during the reaction with fuel gas containing Hg vapour at 204°C, a uniform alloy is formed in the Pd/Al2O3 sorbent, whereas particles containing predominantly a Pd core and Hg-Pd alloy at the periphery are formed in the Pd/SiO2 sorbent. This information has proven beneficial in developing a better understanding of pollutant removal with sorbent material and in particular the effect of Pd particle size in determining the Pd-Hg interaction.
Acknowledgements
GS thanks Royal Society for an Industrial Fellowship and Johnson Matthey plc for hosting the fellowship.
Principal publication and authors
S. Poulston (a), T.I. Hyde (a), H. Hamilton (a), O. Mathon (c), C. Prestipino (c), G. Sankar (a,b) and A.W.J. Smith (a), Phys. Chem. Chem. Phys. 12, 484 (2010).
(a) Johnson Matthey Technology Centre, Sonning Common, Reading (UK)
(b) Department of Chemistry, University College London (UK)
(c) ESRF
References
[1] S. Poulston, E.J. Granite, H.W. Pennline, C.R. Myers, D.C. Stanko, H. Hamilton, L. Rowsell, A.W.J. Smith, T. Ilkenhans and W. Chu, Fuel 86, 2201 (2007).



