- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Dynamics and extreme conditions
- Melting of peridotite to 140 Gpa
Melting of peridotite to 140 Gpa
The solid mantle lies at 2900 km under our feet, and is made up essentially of oxides of magnesium, iron and silicon. It lies above the Earth’s core, which mainly consists of liquid iron. With a pressure of around 1.4 million times the atmospheric pressure and a temperature of more than 4000 Kelvin, the solid mantle is the home for chemical reactions and changes in states of matter that are not completely understood. Seismologists have acknowledged an abrupt reduction of the speed of the seismic waves, which sometimes reaches 30% when getting close to the liquid core boundary. They called these regions ultra-low velocity zones or ULVZs. For the last 15 years, these observations have led scientists to formulate the hypothesis of partial melting of the Earth’s mantle at the core-mantle boundary [1], but, until today, experimental validation at the appropriate temperatures and pressures has remained a challenge.
 |
|
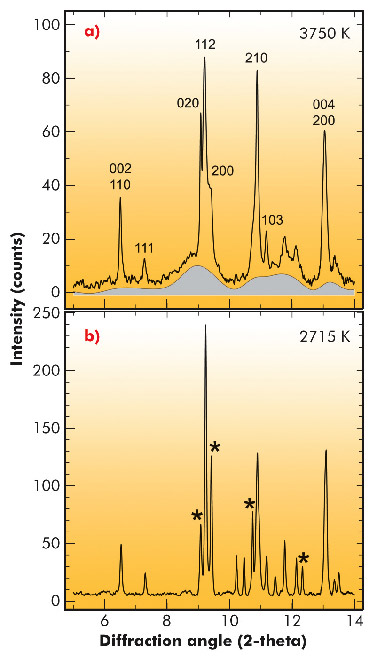
Fig. 11: Diffraction patterns collected at 61 GPa after normalised reference background subtraction: a) above solidus at 3750 K. The diffuse scattering liquid contribution is shown by the shaded area; it does not correspond to a physical structural model of the liquid. HKL indexes are given for remaining diffraction peaks that can be assigned to magnesium silicate perovskite, observed above the solidus temperature at this pressure. b) Subsolidus at 2715 K. Stars denote diffraction peaks of Ca-perovskite and ferropericlase affected by partial melting at these conditions. |
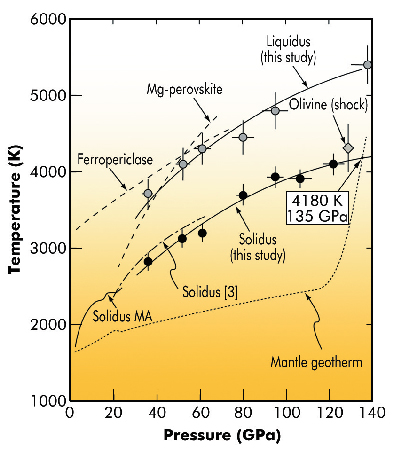
To check the hypothesis of the presence of a partial melt at the base of the mantle, we performed melting experiments on a natural peridotite composition (peridotite is the dominant rock of the Earth's mantle above a depth of about 400 km; below that depth, olivine is converted to denser higher-pressure mineral phases). These experiments were carried out over a range of lower mantle pressures between 36 and 140 GPa, using a laser-heated diamond-anvil cell (DAC) coupled with in situ synchrotron measurements at the high-pressure beamline ID27. To ensure chemical homogeneity at the smallest scale and Fe as mostly Fe2+, a glass was prepared from the peridotite starting material using an aerodynamic levitation device coupled with CO2 laser heating, before being pressurised and heated in a laser-heated diamond-anvil cell. We then used in situ X-ray diffraction as the primary criterion for melting and to determine the order in which crystalline phases melt (Figure 11). From this we could establish, without any extrapolation, fusion curves of the deep Earth’s mantle. We show that partial melting at the base of the mantle is possible when the temperature approaches 4200 K (Figure 12).
 |
|
Fig. 12: Solidus (black solid circles) and liquidus (grey solid circles) of peridotite, along with melting curve data from the literature. Shown are the average estimate of the solidus for KLB-1 peridotite from previous multi-anvil work and melting data for a pyrolitic composition ([3], dot-dashed line). The solid grey diamond indicates the shock melting point of olivine. Melting temperatures of the major lower mantle phases Mg-perovskite and ferropericlase are indicated by long-dashed lines. Our solidus line, extrapolated to the core mantle boundary (solid line), intersects the Earth’s mantle temperature profile (or geotherm) at 135 GPa and 4180 K. |
This observation reinforces the hypothesis of the presence of a deep magma ocean on the top of Earth’s core, which could be as thick as 50 km and explain the presence of ULVZs shown by seismology. It is also attractive to link these observations with an episode of extensive melting that probably affected the primitive Earth, leading to the formation of a deep magma ocean. If the evolution of a terrestrial magma ocean resulted in the formation of a layer of melt at the base of the mantle early in the Earth’s history, our experiments show that some relics of this early magma ocean may have survived at the core mantle boundary. We could also prove that the liquid produced during this partial melting is dense and that it can hold multiple chemical elements, among which are important markers of the dynamics of the Earth’s mantle. Such a layer would thus be an ideal candidate for an unsampled geochemical reservoir hosting a variety of incompatible species, notably the planet’s missing quota of heat-producing elements [2].
Principal publication and authors
G. Fiquet (a), A.L. Auzende (a), J. Siebert (a), A. Corgne (b), H. Bureau (a), H. Ozawa (a,c) and G. Garbarino (d), Science 329, 1516-1518 (2010).
(a) Institut de Minéralogie et de Physique des Milieux Condensés (IMPMC), Université Pierre et Marie Curie, CNRS, Université Paris Diderot, Institut de Physique du Globe de Paris (France)
(b) Institut de Physique du Globe de Paris (France) and Observatoire Midi-Pyrénées, Toulouse (France)
(c) Department of Earth and Planetary Sciences, Tokyo Institute of Technology (Japan)
(d) ESRF
References
[1] Q. Williams and E.J. Garnero, Science 273, 1528 (1996).
[2] M. Boyet and R.W. Carlson, Science 309, 576 (2005).
[3] A. Zerr, A. Diegeler and R. Boehler, Science 281, 243 (1998).



