- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2009
- Electronic structure and magnetism
- Correlation between I-Ag distance and ionic conductivity in AgI-doped fast-ion-conducting glasses
Correlation between I-Ag distance and ionic conductivity in AgI-doped fast-ion-conducting glasses
Fast-ion-conducting (FIC) glasses, whose values of ionic conductivity are comparable to those of liquid electrolytes, are of wide interest for their potential technological applications. Understanding the transport properties in FIC glasses is a challenging problem whose solution may allow one to design new glasses with optimised properties for various applications. Much experimental and theoretical effort has led to some general rules and empirical relations for high ionic conductivity, but a transport model based on local structure is not yet widely accepted [1].
AgI-doped glasses are the best conducting among FIC oxide glasses, with room-temperature conductivities approaching 10-1 ![]() -1cm-1. Although the influence of the AgI doping salt on the glass structure and on the ionic conductivity has been widely investigated, leading to some commonly accepted ideas, a full connection between local structure and ionic conductivity is still lacking. To this aim, EXAFS spectroscopy is particularly well suited, thanks to the selectivity of atomic species, and to the high sensitivity to details of short-range structure.
-1cm-1. Although the influence of the AgI doping salt on the glass structure and on the ionic conductivity has been widely investigated, leading to some commonly accepted ideas, a full connection between local structure and ionic conductivity is still lacking. To this aim, EXAFS spectroscopy is particularly well suited, thanks to the selectivity of atomic species, and to the high sensitivity to details of short-range structure.
An EXAFS investigation of the local structure around iodine was made in different families of AgI-based glasses (borates, phosphates, molybdates, tungstates) affected by different values of ionic conductivity. EXAFS measurements at the K edge of iodine were performed, at liquid nitrogen temperature, at beamline BM29. Crystalline ß-AgI was also measured as reference.
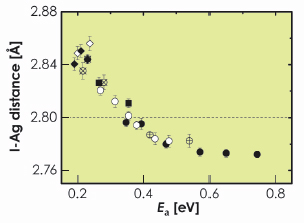
From a phenomenological point of view, the dc ionic conductivity follows an Arrhenius law ![]() T = A exp(– Ea / kBT), where the activation energy Ea is the mean energy required for a cation jump. EXAFS results clearly show that a general correlation exists between the I-Ag bond distance and the activation energy Ea. Figure 99 shows the I-Ag distance obtained from EXAFS for the different glasses examined, plotted against the corresponding activation energy for dc ionic conductivity. The I-Ag distance progressively increases from 2.77 Å in glasses with low ionic conductivity (i.e. high activation energy and low AgI content), to about 2.85 Å in glasses with high ionic conductivity (i.e. low activation energy and high AgI content). Remarkably, glasses with activation energy lower than about 0.35 eV display longer I-Ag distances than crystalline ß-AgI, and vice versa.
T = A exp(– Ea / kBT), where the activation energy Ea is the mean energy required for a cation jump. EXAFS results clearly show that a general correlation exists between the I-Ag bond distance and the activation energy Ea. Figure 99 shows the I-Ag distance obtained from EXAFS for the different glasses examined, plotted against the corresponding activation energy for dc ionic conductivity. The I-Ag distance progressively increases from 2.77 Å in glasses with low ionic conductivity (i.e. high activation energy and low AgI content), to about 2.85 Å in glasses with high ionic conductivity (i.e. low activation energy and high AgI content). Remarkably, glasses with activation energy lower than about 0.35 eV display longer I-Ag distances than crystalline ß-AgI, and vice versa.
 |
|
Fig. 99: I-Ag distance measured by EXAFS in different families of AgI-based glasses at LN temperature, plotted against the activation energy for dc ionic conductivity. The symbols refer to the glass family: |
These results indicate that the I-Ag distance is modified in parallel with the conductivity enhancement of AgI-doped FIC glasses, showing a behaviour that is common to glasses with very different matrices. In agreement with recent works [1, 2], we attribute the expansion of the I-Ag distance to the progressive increase of the number of Ag ions with mixed oxygen and iodine coordination, the strength of the Ag-O bond favouring the expansion of the I-Ag bond.
The activation energy can be written as a sum of two terms: the first one (binding energy) is the mean energy a cation requires to leave its site, the second one (strain energy) is the mean kinetic energy a cation needs to open a ‘‘doorway’’ in the structure to pass through. The decrease of the activation energy induced by salt doping was up to now associated to a lowering of the strain energy [2]. The EXAFS result (i.e., the decrease of the activation energy with increase of the I-Ag distance), suggests that also the binding energy term gives a significant contribution on the enhancement of the ionic conductivity.
References
[1] P. Mustarelli, C. Tommasi, and A. Magistris, J. Phys.Chem. B 109, 17417 (2005), and references therein.
[2] S. Adams and J. Swenson, Phys. Rev. Lett. 84, 4144 (2000).
Principal publication and authors
A. Sanson (a,b), F. Rocca (a), C. Armellini (a), G. Dalba (c), P. Fornasini (c) and R. Grisenti (c), Physical Review Letters 101, 155901 (2008).
(a) CNR-IFN and FBK-CeFSA, Trento (Italy)
(b) Department of Computer Science, University of Verona (Italy)
(c) Department of Physics, University of Trento (Italy)



