- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2009
- Structural biology
- DNA Damage? Call 9-1-1
DNA Damage? Call 9-1-1
Eukaryotic cells respond to DNA damage through a coordinated response in which repair systems are recruited, and the cell cycle arrested, until the damage has been rectified or terminated by apoptosis if too extreme. One such DNA damage sensor is the multifunctional heterotrimeric DNA sliding clamp complex, Rad9-Rad1-Hus1 (9-1-1). In response to DNA damage, 9-1-1 is loaded onto DNA by a specialised form of the replication factor C (RFC) complex. Cell-cycle signalling is then mediated by multiple complexes, specifically the recruitment/stimulation of the checkpoint kinase ATR/ATRIP complex, and ultimately Chk1. 9-1-1 may also play a direct role in DNA damage repair via interaction with a number of DNA repair client enzymes including ‘long-patch’ (or ‘PCNA-scaffolded’) DNA base excision repair components and trans-lesion polymerases.
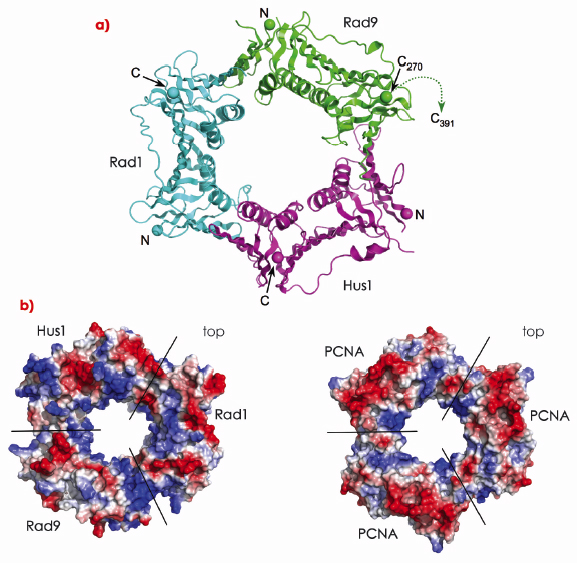
The crystal structure of the human 9-1-1 complex at 2.9 Å resolution was determined using data collected on ID14-1 and ID29. As previously suggested, 9-1-1 has a toroidal architecture similar to PCNA and other DNA clamps (Figure 111a). The 9-1-1 ring encompasses an elliptical hole of sufficient size to accommodate a DNA duplex as experimentally observed for the bacterial ß-clamp. The individual subunits have the same secondary structure topology as other DNA clamps with the outside of the ring formed by faces of ß-sheets, and the hole, lined by ![]() -helices, presenting a basic inner surface to interact with the DNA backbone (Figure 111b).
-helices, presenting a basic inner surface to interact with the DNA backbone (Figure 111b).
 |
|
Fig. 111: Structure of the Rad9-Rad1-Hus1 complex. a) Secondary structure cartoon of the 9-1-1 ring, coloured: Rad9-green, Rad1-cyan and Hus1-magenta. The unstructured ~120 residue C-terminal tail of Rad9 is marked. b) Electrostatic surfaces of 9-1-1 (left) and PCNA (right). Both clamps exhibit strong positive potential (blue) around the central channel through which DNA passes. |
As a heterotrimer, 9-1-1 has a chiral arrangement, with Rad9, Rad1 and Hus1 in anti-clockwise order as viewed from the ‘top’ (studies of PCNA have defined the ‘top’ being the face that provides the binding sites for client enzymes) (Figure 111b). Inspection of the intermolecular subunit interfaces shows clear sterical and chemical specificity for this chiral arrangement.
The 9-1-1 subunits also show substantial differences in structure, particularly in the region that in PCNA provides the typical binding site for ligand proteins (Figure 111a) [1]. Classically, proteins binding to PCNA contain a ‘PIP’ box motif and involve the insertion of three hydrophobic side chains on a short helical segment into a pocket on PCNA. Ligand residues C-terminal to this hydrophobic PIP-box motif may also form an anti-parallel ß-sheet interaction with the inter-domain linker (IDL) connecting the two lobes of the PCNA monomer. The IDL segments in all three 9-1-1 subunits are well ordered and exhibit different conformations (Figure 111a). While Rad9 could accommodate a hydrophobic PIP-box motif, this binding pocket is completely blocked in Rad1, and partially blocked in Hus1.
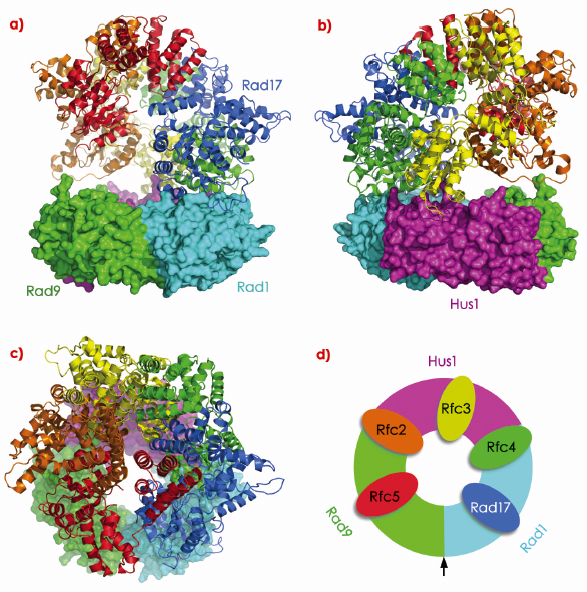
Like PCNA, 9-1-1 is loaded onto DNA via a ring-open intermediate in an ATP-dependent mechanism mediated by the specialised Rad-RFC complex where the Rfc1 subunit is replaced by Rad17. Crystallographic analysis of a yeast RFC-PCNA complex in the presence of ATP![]() S [2] reveals a complex pattern of interactions between the RFC complex and hydrophobic PIP-box binding pockets on PCNA. With the determined structure of the 9-1-1 IDLs, and availability of the hydrophobic PIP-box binding pockets in each subunit, it is possible to model a 9-1-1-Rad17-RFC complex (Figure 112). This maps the biochemically predetermined break point in PCNA to the Rad9-Rad1 interface in 9-1-1, the least substantial of the three interfaces – burying only ~1100 Å2, and is consistent with biochemical data.
S [2] reveals a complex pattern of interactions between the RFC complex and hydrophobic PIP-box binding pockets on PCNA. With the determined structure of the 9-1-1 IDLs, and availability of the hydrophobic PIP-box binding pockets in each subunit, it is possible to model a 9-1-1-Rad17-RFC complex (Figure 112). This maps the biochemically predetermined break point in PCNA to the Rad9-Rad1 interface in 9-1-1, the least substantial of the three interfaces – burying only ~1100 Å2, and is consistent with biochemical data.
 |
|
Fig. 112: Clamp loader interactions with 9-1-1. a) Model of a 9-1-1-Rad17-RFC complex. In this model, Rad9 and Hus1, interact with the Rfc2,3,4 and 5 subunits, while Rad1 presents a distinct interaction surface to interact with the 9-1-1-specific Rad17 subunit. b) As a) but rotated 180º around the vertical. c) As a) but rotated 90º around the horizontal. d) Schematic of the modelled interactions between Rad17-RFC and 9-1-1. The arrow denotes the predicted 9-1-1 “break-point” for DNA loading. |
Finally, we found that interaction of ligand proteins with 9-1-1 appears to be regulated by genotoxic stress, with the cell cycle regulator p21cip1/waf1 binding to 9-1-1 in competition with the LP-BER component FEN1. Unlike PCNA, which as a homotrimer possesses three equivalent binding sites, there appears to be a single site on the 9-1-1 heterotrimer to which FEN1 and p21cip1/waf1 bind in competition.
The crystal structure of 9-1-1 provides a clear molecular view of this multifunctional checkpoint clamp revealing significant differences between the three subunits. In particular, the PIP-box binding pocket which provides the main binding site for ligand proteins, is substantially altered in all the subunits of 9-1-1 with implications for clamp loading via the Rad17-RFC complex and regulation through cell cycle regulators such as p21cip1/waf1.
References
[1] J.M. Gulbis, Z. Kelman, J. Hurwitz, M. O’Donnell and J. Kuriyan, Cell, 87, 297-306 (1996).
[2] G.D. Bowman, M. O’Donnell and J. Kuriyan, Nature, 429, 724-730 (2004).
Principal publication and authors
A.S. Doré (a), M.L. Kilkenny (a), N.J. Rzechorzek (a,b) and L.H. Pearl (a), Mol. Cell, 34 (6), 735-745 (2009).
(a) CR-UK DNA Repair Enzymes Group, Section of Structural Biology, The Institute of Cancer Research, London (UK)
(b) Department of Biochemistry, University of Cambridge (UK)



