- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2008
- Structural biology
Structural biology
Introduction
Macromolecular crystallography has continued to evolve at the ESRF. The investment made in automation and beamline reliability is bearing fruit in a number of interesting ways. It is now clear that, in many cases, the complexity of the biological systems under study can only be fully revealed by combining extensive initial sample evaluation with detailed examination of the most promising crystals on the beamline best suited to the experiment. Obtaining the maximum information from a crystal also requires optimisation of the data collection procedure. Only by employing more complex experimental strategies can we hope to obtain a successful outcome to large numbers of experiments.
The improved operational reliability of the macromolecular crystallography (MX) beamlines has enabled a recent change to the way they can be accessed. Remote access was introduced during 2008 and allows complete instrument control from the user’s home laboratory. This mode of access has already proven popular, currently accounting for more than 20% of all macromolecular crystallography experiments, a figure we expect to see rising. However, the advent of remote access has revealed a number of areas where further instrument development would be useful. It has also emphasised the need for the ESRF to adapt staffing levels and staff profiles in order to fully support such access to the beamlines.
The results presented in this chapter reveal the extent to which access to a broad portfolio of beamlines and synchrotron techniques is important for achieving success. We observe that both microfocus X-ray beams and tunability of the X-ray source are critical. In this latter context, the CRG beamlines BM14, BM16 and BM30A also play an important role, providing excellent services for their respective communities. The increased study of macromolecular complexes has led to a resurgence of interest in small-angle X-ray scattering from protein solutions. To provide much needed extra capacity ID14-3 has been converted to a SAXS beamline in a collaboration with the EMBL; it will provide rapid access for experiments in this area. The first such experiments were carried out on the beamline in 2008 and we look forward to a productive year in 2009.
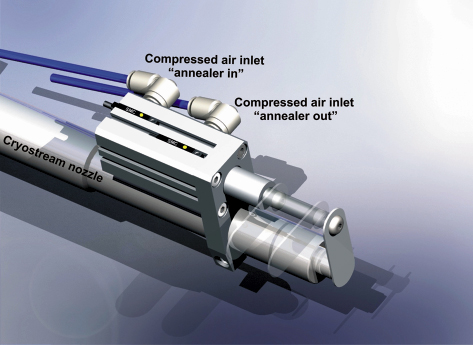
Some of the fruits of the research undertaken in 2008 on the ESRF MX beamlines are reported in this chapter. We concentrate only on biological structures obtained and the insights that these give. Methodology contributing to this success is reported in the chapter on Methods and Instrumentation, and in Figure 63, which presents a new annealing device available on the beamlines.
 |
|
Fig. 63: Cryogenic cooling is used to prolong the life-time of crystals in an X-ray beam. However, in some cases flash-cooling degrades crystal quality if conditions are not ideal. Annealing or tempering of samples adversely affected by cryocooling can often improve diffraction quality and is now considered a standard technique in macromolecular crystallography. A small, inexpensive automatically-operated annealing device is installed on all ESRF macromolecular crystallography beamlines that allows annealing of samples either from the beamline or by users accessing the beamline remotely. For details see: T. Giraud et al., J. Appl. Cryst. 42, 125 (2009). |
The current degree of automation available on the MX beamlines is due to the consistent hard work of the ESRF BLISS, SciSoft, and MX Groups in partnership with the EMBL Grenoble outstation. Their efforts were recognised by the annual BESSY innovation award. Work on renewing the MX suite of beamlines will continue through 2009 with the development of detailed plans for new beamlines and modes of access. We look forward to the help and input of our User community in the continuation of these projects.
G. Leonard and S. McSweeney



