- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2008
- Soft condensed matter
- Probing protein structural changes in solution
Probing protein structural changes in solution
Proteins are sequences of aminoacids that fold in 3D structures to accomplish their biological functions. Several studies have shown that protein conformational changes often play a crucial role in function regulation. So far, direct structural information on molecular motions has been obtained only in the crystal state by time-resolved protein crystallography [1]. However, protein crystals, while allowing model-free atomic resolution reconstruction, may induce stress/strain in the protein and hinder biologically-important structural changes. For proteins in solution, only indirect information has been obtained so far through time-resolved optical spectroscopy [2], thus highlighting the need of more direct, time-resolved, structural probes. In the last decade, protein solution scattering has undergone tremendous improvements in instrumentation and understanding of the experimental data, thus opening the possibility of studying protein structures in solution although at somewhat limited resolution of 5-10 angstroms [3].
We have therefore attempted to combine the exceptional time resolution achievable at ID09B (down to 100 ps) with the structural sensitivity already demonstrated for protein solution scattering.
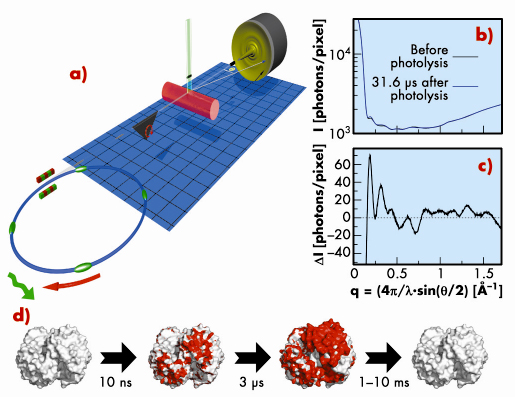
We demonstrate this approach by using it to monitor the structural changes of different proteins after photodissociation of the ligand. The basic idea of the experiment is as follows: X-ray pulses are selected from the storage ring using a synchronised ultra-fast mechanical shutter (Figure 59). Synchronised laser pulses are produced by the laser system available at ID09B, where different pulse lengths and laser wavelength can be used. The laser/X-ray pulse pairs (suitably shifted by a time delay that can be varied electronically with an accuracy of ~ 10 ps) are sent to the sample to excite and probe the structure respectively. Typically, for a given time delay, the sample has to be exposed to about 1000 single laser/X-ray pulses to make efficient use of the CCD dynamic range.
 |
|
Fig. 59: Schematic of the TR-WAXS experimental technique (Adapted by permission from Macmillan Publishers Ltd: Nature Methods M. Cammarata et al., Nature Methods 5, 881, copyright (2008)). |
For reciprocal vector (q = 4![]() /
/ ![]() sin(
sin(![]() /2)) in the range 0.05 to 2 Ang-1 protein structural features gives rise to modulations of the scattering pattern that are fingerprints of the protein structure thus giving a direct structural probe. Since the light-induced differences are relatively small (< 5%), the data are usually presented as laseron-laseroff difference scattering patterns.
/2)) in the range 0.05 to 2 Ang-1 protein structural features gives rise to modulations of the scattering pattern that are fingerprints of the protein structure thus giving a direct structural probe. Since the light-induced differences are relatively small (< 5%), the data are usually presented as laseron-laseroff difference scattering patterns.
To test this new experimental approach we studied human haemoglobin because it offers a well-known example of structurally tuned biological function. Haemoglobin is made of four subunits each of them able to reversibly bind small molecules such as oxygen or carbon monoxide; it performs its biological task efficiently (oxygen delivery) thanks to the equilibrium between two conformations (called “R” and “T”) having a dramatically different ligand affinity. Because of its fundamental biological importance, the transition between the R and T conformations (called “quaternary transition”) has attracted the interest of many researchers over the last decades.
In our experiment, a short laser pulse was able to break the protein-ligand bonds thus triggering protein structural relaxation from the initial ligand bound (R) structure to the unbound (T) structure; ligand rebinding from the solution eventually restores the initial condition.
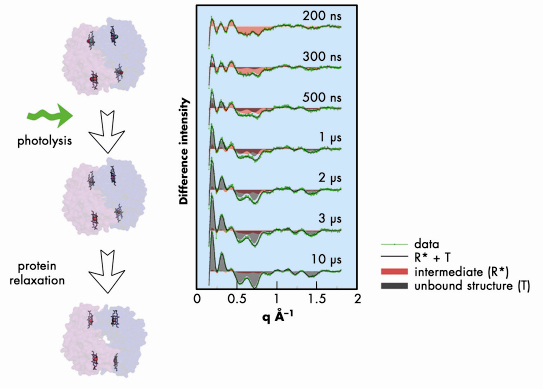
Thanks to the high photon flux available at ID09B, we were able to provide a direct observation of the R –> T transition and to measure unambiguously the “waiting time” before the quaternary transition takes place. We found a time scale of about 2 µs challenging the previous assignment obtained with absorption spectroscopy [4] but in agreement with more recent time-resolved Raman data [2]. The main results are shown in Figure 60. The difference scattering patterns (laser at a given time delay – laser off) show a clear evolution of the signal that can be interpreted as the transition from an intermediate structure to the unbound equilibrium structure. The fact that the 3 µs and the 10 µs curves are very similar immediately suggests a time scale for the transition of a few microseconds.
 |
|
Fig. 60: Experiment on haemoglobin: left panels show the schematic of the flash photolysis, right panels show the data at different time delays (green) along with the reconstruction using two time delays (black). |
This study shows that time resolved scattering can be a valuable experimental technique to gain insight into protein structures and their changes. Different excitation mechanisms like pH jump or T-jump could also be used and preliminary tests are being carried out.
Principal publication and authors
M. Cammarata (a,b), M. Levantino (c), F. Schotte (d), P.A. Anfinrud (d), F. Ewald (a), J. Choi (e), A. Cupane (c), M. Wulff (a), H. Ihee (e), Nature Methods 5, 881 (2008).
(a) ESRF
(b) Centre for Molecular Movies, Niels Bohr Institute, University of Copenhagen (Denmark)
(c) Department of Physical and Astronomical Sciences, University of Palermo (Italy)
(d) Laboratory of Chemical Physics, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda (USA)
(e) Center for Time-Resolved Diffraction, Department of Chemistry (BK21), Korea Advanced Institute of Science and Technology, Daejeon (Republic of Korea)
References
[1] F. Schotte, M.H. Lim, T.A. Jackson, A.V. Smirnov, J. Soman, J.S. Olson, G.N. Phillips, M. Wulff, P.A. Anfinrud, Science 300, 1944 (2003).
[2] G. Balakrishnan, M.A. Case, A. Pevsner, X.Z.C. Tengroth, G.L. McLendon, T.G. Spiro, J. Mol. Biol. 340, 843 (2004).
[3] D.I. Svergun and M.H. Koch, Rep. Prog. Phys. 66, 1735 (2003).
[4] J. Hofrichter, J.H. Sommer, E.R. Henry, W.A. Eaton, Proc. Natl. Acad. Sci. USA 80, 2235 (1983).



