- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2008
- High resolution and resonance scattering
- Visualising the ignition of catalytic partial oxidation of methane
Visualising the ignition of catalytic partial oxidation of methane
Catalytic reactions are present in many chemical processes. More than 90% of all chemical products have undergone at least one catalytic step. The development of new catalysts has often been triggered by the development of new techniques. Especially important is energy-related catalysis, to make catalysts more efficient and more stable and to substitute fossil fuels, e.g. by biomass-related processes. One important reaction is the catalytic partial oxidation (CPO) of methane for the production of synthesis gas, which is catalysed by noble metals such as Pt, Rh and Ir. It can be regarded a key step in transformation of natural gas or biomass into liquid fuels such as methanol and higher alkanes [1,2]. Changes in chemical reactions like the CPO with time and/or in space are often correlated to a change in state and structure of the catalyst [3]. To gain deeper insight, it is desirable to follow such structural changes in a spatiotemporal manner.
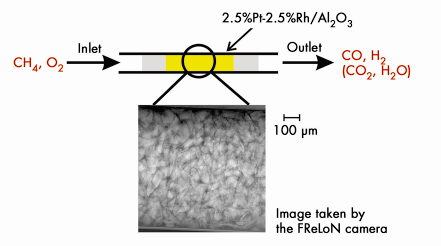
We studied the ignition of the catalytic partial oxidation of methane on a Pt-Rh/Al2O3 catalyst by spatially and time-resolved imaging of a catalytic reactor using a FReLoN (fast readout low noise) camera behind the microreactor [4]. The setup is shown in Figure 16.
 |
|
Fig. 16: Schematic figure of the catalytic microreactor for partial oxidation of methane to CO and H2 on a 2.5wt%Pt-2.5wt%Rh/Al2O3 catalyst; a 1 mm x 1 mm snapshot taken with the FReLoN camera is depicted below. |
The oxidation state of the noble metals is strongly dependent on the reaction conditions. Below the ignition temperature of the reaction towards CO and H2, the catalyst is in an oxidised state, whereas above the ignition temperature, a reduction of the noble metals occurs. This can be followed by X-ray absorption spectroscopy (see e.g. ref. [3]). In particular, a strong change in X-ray absorption occurs at the whiteline of platinum (11568 eV). By tuning the energy of the incident X-rays to the maximum of the whiteline, a variation in X-ray absorption can uncover where the platinum constituent is reduced. In fact, by collecting X-ray transmission images of the microreactor with a frame rate of 4s–1, a strong structural change in X-ray absorption, and thus the structure of the catalyst, could be found during the ignition of the CPO of methane. Selected images at times t1 (Pt is still in an oxidised state over the whole catalyst bed) and t > t1 are depicted in Figure 17. Reddish and violet colours indicate a lower X-ray absorption and thus reduced Pt-species.
 |
|
Fig. 17: a) to e): Images recorded at the whiteline energy of Pt (Pt L3-edge; 11586 eV) as a function of time with flow conditions as depicted in Figure 16: reddish colour indicates the formation of reduced Pt-species; f) X-ray absorption image recorded below the CPO ignition temperature, showing that the images were taken at the end of the catalyst bed composed of Pt-Rh/Al2O3 on the left and only inert alumina material on the right. |
Images 2a) to 2e) were obtained by subtraction of the X-ray absorption image at time t from Figure 17f) below the ignition, which emphasises the difference in absorption and corrects for inhomogeneities in the sample. The full series taken of the ignition process was compiled into a movie and we observed that the front of reduced platinum species moves from the outlet towards the inlet of the reactor. The observed reduction of the noble metal occurred at the same time as the chemical reaction ignited (detected by on-line mass spectrometry). Note that the ignition starts at some specific single particles in the catalytic reactor (compare Figures 17b and 17c).
The movement of the front from the outlet towards the inlet can be explained with a total oxidation - reforming mechanism. Below the ignition temperature methane is fully combusted to carbon dioxide and water over the whole length of the catalyst bed. Upon heating the reaction self-accelerates on some of the catalyst grains until all oxygen is consumed and then some new metallic species are formed. These new species promote the activation of methane and thus lead to the formation of hydrogen and carbon monoxide. Furthermore, the methane oxidation accelerates and the reduction propagates towards the inlet of the reactor, where the total oxidation of methane still occurs over oxidised noble metal particles.
In conclusion, by using high resolution transmission X-ray imaging we could determine the structure of a catalyst not only in situ but also in a spatiotemporally resolved manner. Thereby, important insight into the first stages of the ignition reaction could be gained. This allows an understanding of the ignition/ extinction of catalytic reactions, reaction mechanisms and also chemical oscillations in more detail.
Principal publication and authors
B. Kimmerle (a), J.-D. Grunwaldt (a,b), A. Baiker (a), P. Glatzel (c), P. Boye (d), S. Stephan (d) and C.G. Schroer (d), J. Phys. Chem. C, DOI:10.1021/jp810319v (2009).
(a) Institute for Chemical and Bioengineering, Department of Chemistry and Applied Biosciences, ETH Zürich (Switzerland)
(b) Department of Chemical and Biochemical Engineering, Technical University of Denmark (Denmark)
(c) ESRF
(d) Institute of Structural Physics, TU Dresden (Germany)
References
[1] D.A. Hickman, and L.D. Schmidt, Science 259, 343 (1993).
[2] J.-D. Grunwaldt, L. Basini, B.S. Clausen, J. Catal. 200, 321 (2001).
[3] J.-D. Grunwaldt, S. Hannemann, C.G.Schroer, A. Baiker, J. Phys. Chem. B 110, 8674 (2006).
[4] J.-D. Grunwaldt, B. Kimmerle, A. Baiker, P. Boye, C.G. Schroer, P. Glatzel, C. Borca, F. Beckmann, Catal. Today, on-line, doi: 10.1016/j.cattod.2008.11.002.



