- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2007
- X-ray Imaging and Optics
- Micrometric atypical pigment identified in Grünewald Isenheim Altarpiece
Micrometric atypical pigment identified in Grünewald Isenheim Altarpiece
Mathias Grünewald is an important German Renaissance painter. He created his magnum opus, the Isenheim Altarpiece, during ~1512–1516. His renown is mainly based on his unique skill in handling colours. In preparation for an exhibition held at Colmar (France) and Karlsruhe (Germany) late 2007, chemical analyses were conducted to establish the list of painting pigments used by the artist. In a first approach, non-invasive analytical portable instruments, allowing in situ screening, were favoured. Interestingly, antimony was detected in several parts of the artwork, in particular for the rendering of metallic reflects, as shown in Figure 131 (sleeping guard in the Resurrection scene). Although sulphur seemed to be associated with antimony, this correlation was questionable due to the high concentration of lead that strongly overlaps with sulphur emission lines. Antimony is not commonly found in paintings. It is usually associated with Naples yellow, a lead antimony oxide, used in paintings in a much later period.
 |
|
Fig. 131: Part of the Resurrection, from the Isenheim Altarpiece, by Grünewald (picture E. Lambert, © C2RMF). The white star indicates where the micro-sampling was done. |
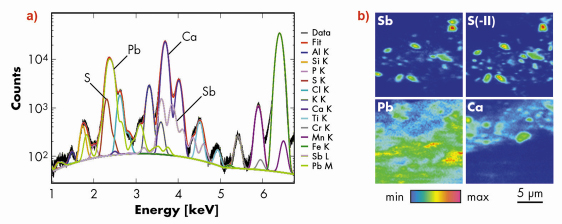
To further the identification of Grünewald’s antimony pigment, micro-sampling was performed, guided by portable XRF measurements. When such micro-sampling are allowed on ancient paintings, fragments are usually tiny, precious, heterogeneous and complex, which makes their chemical analysis arduous. As illustrated here, micro-imaging techniques are particularly relevant as they can simultaneously provide identification and localisation of the various ingredients, even in intricate mixtures. Transversal cross-sections were prepared to reveal the whole stratigraphy of the painting fragments. Micro X-ray fluorescence was carried out at the ID21 beamline, first at the antimony L-edges and then at the sulphur K-edge. Fitting of fluorescence data was arduous due to interferences of K-lines of sulphur with M-lines of lead, and of K-lines of calcium with L-lines of antimony (Figure 132a). This data processing was performed using PyMCA software developed at the ESRF [1]. Elemental maps show that antimony distribution is limited to an intermediate layer, above a lead layer and below a varnish superficial layer (Figure 132b). They also reveal that antimony and sulphur distributions are similar and that these elements are present as few micrometres-sized grains, surrounded by a lead white matrix. These maps suggested the probable presence of stibnite (Sb2S3), a grey antimony sulfide. Additional µ-XANES was performed at the Sb LIII-edge and the S K-edge to confirm this assumption and to refute the presence of any other antimony pigments that can be used in painting (in particular Naples yellow (Pb2Sb2O7) or Kermesite (Sb2S2O)). Special care was given to minimise the micro-probe drift while scanning the energy. Indeed, micro-probe stability was a critical issue to selectively analyse the antimony grains without shifting into the lead white matrix. Sulphur was found to be in a reduced state (sulphide), without chemical bonding to lead. Comparison with reference compounds gave clear support to the suggestion that the antimony-containing compound was stibnite. This pigment has been found in only about ten other paintings, all of them originating from Italy in the first half of the 16th century. Grünewald tailored the metallic aspects of the painting by mixing micrometric stibnite grains with a lead white matrix.
 |
|
Fig. 132: Identification of stibnite micrometric grains by µ-XRF: a) fluorescence spectrum obtained at 7.2 keV, over the cross-section and fitted with PyMCA. b) fitted elemental mapping of antimony, sulfides, lead and calcium. Map size: 17 x 16 µm2; step size: 0.25 x 0.25 µm2; beam size: 1 x 0.4 µm2 (hor. x ver.). |
Lateral resolution, chemical sensitivity, and imaging are the key points that explain the advantage of synchrotron-based imaging techniques for the analysis of fragments of ancient paintings. The continuous development of software is very helpful for the processing of data obtained, not only on synchrotrons but also with laboratory as well as portable instruments. This study illustrates the complementarity between portable and synchrotron instruments in the discovery of the secrets of the ancient painters.
References
[1] V.A. Solé, E. Papillon, M. Cotte, P. Walter, J. Susini, Spectrochim. Acta Part B 62(1), 63-68 (2007).
Principal publication and authors
M. Cotte (a,b), E. Welcomme (a), V.A. Solé (b), M. Salomé (b), M. Menu (a), P. Walter (a), J. Susini (b), Anal. Chem. 79, 6988-6994 (2007).
(a) Centre of Research and Restoration of the French Museums, CNRS-UMR171, Paris (France)
(b) ESRF



