- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2006
- Structural Biology
- Crystal structures of the ribosome in complex with RNAs and release factors
Crystal structures of the ribosome in complex with RNAs and release factors
The ribosome is the large (2.5 Megadalton) protein-RNA complex in the cell that makes proteins according to the sequence of codons (groups of 3 bases) present on the genetic template in the form of mRNA [1]. The process is extremely complicated and involves large tRNA substrates that bring in the amino acids to be added to the protein chain. Ribosomes consist of two subunits, the small (30S) which binds the mRNA template and the large (50S), which contains the active site for peptide bond formation between the incoming amino acid and the growing protein chain. These subunits undergo major conformational changes during translation and the tRNAs and mRNA move precisely through the ribosome in a remarkably precise and rapid process [2].
Following the solution of the atomic structures of both ribosomal subunits in 2000, the focus has shifted to crystallography of the ribosome in complex with its substrates in various states, in an effort to understand the detailed mechanism of translation. This process is difficult because it is hard to purify homogeneous complexes of a defined state that are stable enough to crystallise. Moreover, with such large complexes, most crystals do not diffract well. We have overcome this problem using a variety of techniques.
 |
|
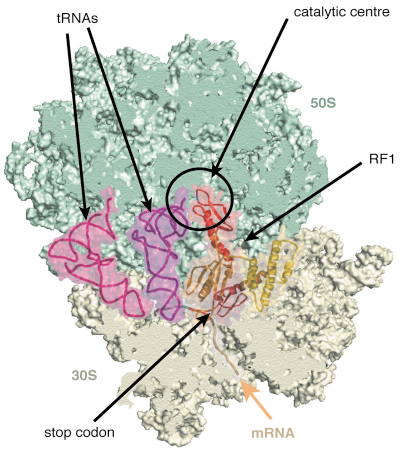
Fig. 66: A view of the structure of the ribosome with mRNA, tRNAs and release factor RF1. |
Release factors RF1 and RF2 recognise when the ribosome has reached a stop codon. In response to this they trigger the release of the finished protein chain from the ribosome. However, it is not currently known how release factors can distinguish between the various stop codons or between them and other codons that code for amino acids. We “tagged” the release factors RF1 and RF2 and used the tag to separate fully assembled ribosomal complexes from defective ones. These complexes were crystallised and data collected at ID14-4 [3]. The structure (Figure 66) shows how a “GGQ” motif present on RF1 and RF2, that is thought to be required for cleavage of the peptide chain, is ordered in the catalytic centre of the 50S subunit. A loop thought to contain the motif that recognises stop codons is close to the position of the codon in the 30S subunit. These structures, at about 6 Å resolution, allowed accurate placement of the domains. We are currently trying to improve the resolution of the crystals so that we can see atomic details of these interactions.
 |
|
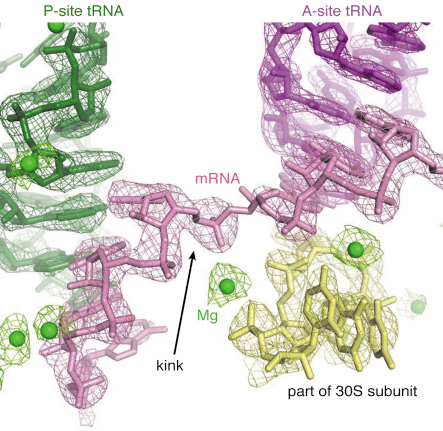
Fig. 67: Detail of a high-resolution structure of the ribosome complexed with mRNA and tRNA, showing electron density for a kink in the mRNA as well as metal ions (green spheres), one of which stabilises the kink. |
Structures of functional complexes of ribosomes have remained at about 5-6 Å resolution for the past few years. At this resolution, it is possible to fit known structures or domains into electron density maps, but it is impossible to interpret unknown regions or reveal details of the chemical interactions in the molecule. We made a concerted effort to obtain improved crystals of the 70S ribosome in complex with mRNA and tRNA. This effort involved screening hundreds of crystals, both at Daresbury Laboratory and at the ID14 beamlines at ESRF, where the automatic crystal mounting robotics greatly facilitated the process. Eventually, we were able to identify an improved crystal form that allowed us initially to collect a 4.6 Å data set at ID14-4 and, after further improvement in crystallisation, a 2.8 Å data set at beamline X10SA at the SLS. This structure (Figure 67) reveals atomic details of how the ribosome interacts with its mRNA and tRNA ligands, and also how metal ions play an important structural and functional role in the ribosome [4]. For example, metal ions are not only important to hold the structure together, they also form crucial elements of the “bridges” between the two subunits. Interestingly, a metal ion also stabilises a kink in the mRNA that helps to demarcate the difference between the sites for the tRNA that holds the growing protein chain (“P site”) and that which brings in the new amino acid (“A site”). We hope that this new crystal form will be useful for studying how various ligands and antibiotics bind to the entire ribosome in a defined state.
References
[1] V. Ramakrishnan, Cell 108, 557-572 (2002).
[2] A movie that outlines this process can be seen at http://alf1.mrc-lmb.cam.ac.uk/~ribo/movies/
[3] S. Petry et al., Cell 123, 1255-66 (2005).
[4] M. Selmer et al., Science 313, 1935-42 (2006).
Principal Publication and Authors
M. Selmer, C.M. Dunham, F.V. Murphy IV, A. Weixlbaumer, S. Petry, A.C. Kelley, J. Weir and V. Ramakrishnan.
MRC Laboratory of Molecular Biology, Cambridge (UK)



