- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2006
- Materials Science
- Carbonia: the amorphous silica-like carbon dioxide
Carbonia: the amorphous silica-like carbon dioxide
Among the group IV elements only carbon forms stable double bonds with oxygen at ambient conditions and, at variance with SiO2 and GeO2, the non molecular single-bonded crystalline form of CO2, phase V, only exists at high pressure [1]. Similarly, the amorphous phases of SiO2 (a-silica) and GeO2 (a-germania) are well known and stable at room conditions, but the amorphous, non molecular, form of CO2 has been described so far only as a result of first-principle simulations [2]. We achieved the synthesis of an amorphous, silica-like form of carbon dioxide, a-CO2 or “a-carbonia”, by compressing the molecular phase III of CO2 above 40 GPa, at temperatures as high as 680 K, in a diamond anvil cell. State-of-the-art infrared absorption, Raman and synchrotron X-ray diffraction measurements were used to assess the amorphous character of the new material. Comparison with vibrational and diffraction data for amorphous silica and germania, as well as with the structure factor calculated on the a-CO2 sample obtained by first-principle molecular dynamics [2], shows that a-carbonia is structurally homologous to the other group-IV dioxide glasses.
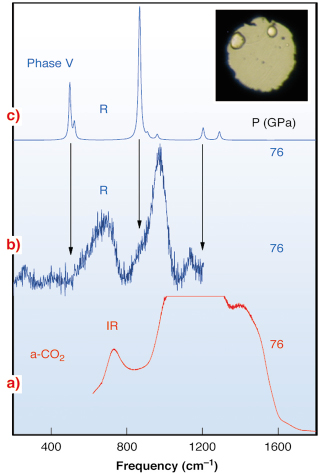
In Figure 30 we report vibrational spectra of temperature quenched a-CO2. The infrared spectrum shows broad absorption bands, whose large width is related to the disorder. By analogy to the infrared spectra a-SiO2, bands of a-carbonia have been assigned to different modes of the carbon–oxygen network made of single C–O bonds. The Raman spectrum also exhibits broad bands, compared to that of crystalline phase V, which in turn has much narrower lines. The Raman lines of CO2-V are close to the a-CO2 band edges, therefore it was speculated that bands of a-CO2 reflect vibrational density-of-state features of CO2-V. This also supported the notion that a-CO2 is the glassy counterpart of phase V.
 |
|
Fig. 30: Vibrational spectra of non-molecular CO2: a) Infrared spectrum of a-carbonia (the absorption of the strongest band is partially saturated); b) and c) Raman spectra of a-carbonia and phase V, respectively. Arrows indicate the correspondence of phase V Raman lines to band edges of a-carbonia. Inset: typical temperature quenched a-CO2 sample (P = 61 GPa, sample diameter about 80 µm; two ruby chips are close to the gasket). |
As a crucial test of structural disorder in the new material, we measured its static structure factor S(Q) (Figure 31). The S(Q) is dominated by a broad peak (A), which lies in the Q region corresponding to the nearest neighbours atomic length scale, and a much broader peak (B) positioned at higher Q. The theoretical S(Q), calculated on a fully tetrahedral atomic structure obtained by first-principle molecular dynamics [2], reproduces both experimental peaks A and B. We also compare the S(Q) of a-carbonia to that of a-SiO2: peaks A of the two materials are close to each other, while differences in the overall profile can mainly be ascribed to the different atomic form factors.
 |
|
Fig. 31: Static structure factor S(Q) of a-CO2. a) and b): computer simulated S(Q) for CO4 (ab initio molecular dynamics) and higher (classical molecular dynamics), up to octahedral, carbon coordination, respectively; c), d) and e): temperature quenched samples. The structure factor of sample (e) has been measured by decreasing pressure. The experimental S(Q) of a-SiO2 is also reported at 42 GPa (dotted line) and 28 GPa (dashed line). Symbols indicate diffraction peaks of: (stars) rhenium gasket, (squares) phase V (the closest line to peak A of a-CO2), and (triangles) phase III. Inset: picture of ab initio simulated a-carbonia. |
We notice that peak A is very close to the strongest Bragg peak of crystalline CO2-V, whose intensity has been suggested to be enhanced by disorder, indicating once again that a-CO2 is the amorphous counterpart of phase V. Upon decreasing pressure below 16 GPa, a-CO2 transforms into a new amorphous material, whose diffraction features appear to be related to the strongest Bragg peaks of phase III, and which we suggest to be the amorphous molecular counterpart of phase III. Below 10 GPa phase I (dry ice) is formed by recrystallisation of the amorphous material.
With the discovery of “a-carbonia”, a new, extended analogy emerges between the structures of CO2 and those of the known SiO2/GeO2 polymorphs. Our findings lead to the general conclusion that the class of archetypal network-forming disordered systems as a-silica, a-germania, a-Si, a-Ge, and water, must be extended to include a-CO2.
References
[1] M. Santoro, and F. A. Gorelli, Chem. Soc. Rev. 5, 918 (2006).
[2] S. Serra, C. Cavazzoni, G. L. Chiarotti, S. Scandolo, and E. Tosatti, Science 284, 788 (1999).
Principal Publication and Authors
M. Santoro (a,b), F.A. Gorelli (a,b), R. Bini (a,c), G. Ruocco (b,d), S. Scandolo (e) and W. Crichton (f), Nature 441, 857 (2006).
(a) LENS, Sesto Fiorentino (Italy)
(b) CRS-SOFT-INFM-CNR, c/o Università di Roma “La Sapienza” (Italy)
(c) Dipartimento di Chimica dell’Università di Firenze, Sesto Fiorentino (Italy)
(d) Dipartimento di Fisica, Università di Roma “La Sapienza” (Italy)
(e) ICTP and CRS-DEMOCRITOS-INFM-CNR, Trieste (Italy)
(f) ESRF



