- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2005
- Materials Science
- View on Grain Nucleation and Growth Kinetics during Solidification
View on Grain Nucleation and Growth Kinetics during Solidification
Understanding the crystallisation process during solidification is an essential step in order to tailor the mechanical properties of solidified materials. The physical processes that govern crystallisation are grain nucleation and the subsequent grain growth [1]. In the industrial production process of aluminium alloys the addition of TiB2 particles along with solute titanium is widely used to enhance the nucleation rate and control the grain growth during solidification [2]. This procedure is generally referred to as grain refinement. Although the mechanisms responsible for grain refinement were studied extensively during the last few decades, a comprehensive understanding is still lacking due to experimental difficulties in monitoring the grain nucleation and growth in situ. A question of particular interest is how added TiB2 particles along with solute titanium results in grain refinement, while no grain refinement is observed if one of the two is missing.
Our present study investigates the nucleation and the growth of individual aluminium grains during solidification. An aluminium alloy with solute titanium (0.1 wt.%) and added TiB2 particles (0.1 wt.%) was chosen as a model system. Time-resolved X-ray diffraction measurements during solidification of this alloy were performed at ID11. The evolution of 2D diffraction images during solidification is shown in Figure 44. Before solidification, the short-range order in the liquid gives rise to diffuse scattering, indicated by two broad rings L1 and L2 on the 2D image. During solidification, we observe the appearance of bright spots, which correspond to Bragg peaks from individual grains of the solid phase that grow within the liquid phase.
 |
|
Fig. 44: X-ray diffraction patterns of the aluminium alloy with solute titanium and added TiB2 particles at different stages of the solidification process during continuous cooling: (a) the liquid phase, (b) the mixed liquid-solid phase, and (c) the solid phase. |
Extraction of quantitative information was performed by monitoring the number and intensity of individual diffraction spots during solidification. By counting the number of spots as a function of time, the evolution of the nucleation rate of the solid aluminium grains was obtained. The scaled intensity variation in the first liquid peak (L1) provides information about the evolution of solid phase fraction. The growth behaviour of individual aluminium grains is determined by continuously monitoring the intensity of the each diffraction spot during solidification.
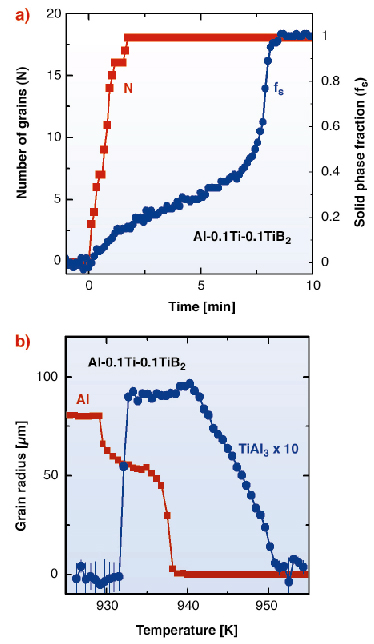
In Figure 45a the time evolution of nucleation process and the solid phase fraction is shown for solidification during continuous cooling at rate of 1 K/min. The results demonstrate that the nucleation process is limited to the initial stage of the solidification and is complete at a solid phase fraction of about 20%. In fact the growth of nucleated grains leads to a significant release of latent heat that limits the undercooling available to activate later nucleation events as the transformation proceeds. Figure 45b shows the evolution of the grain radius of an individual aluminium grain as a function of temperature during solidification. The observed growth behaviour of the aluminium grains is controlled by the diffusion of solute titanium and the release of latent heat. As titanium has a strong affinity for the solid phase, its concentration in the melt decreases as the solidification proceeds. A careful analysis of the measured diffraction patterns shows a limited number of weak diffraction spots, originating from a TiAl3 that appears before the start of the solidification process. In Figure 45b the evolution of an individual TiAl3 grain is shown. The TiAl3 phase is found to form about 10 K above the experimental solidification temperature of aluminium. At the nucleation temperature of the aluminium, the intensity of the TiAl3 grain starts to decrease, and finally vanishes near the end of the transformation. Due to its better nucleation efficiency, the formation of the TiAl3 phase in the presence of both solute titanium and TiB2 particles was long proposed, but due to the meta-stable nature of the TiAl3 phase there was no experimental evidence available [2].
 |
|
Fig. 45: Grain nucleation and growth in the aluminium alloy during solidification. (a) Total number of nucleated grains in reflection and the corresponding solid phase fraction as a function of time. (b) Grain radius of an individual aluminium grain and an individual TiAl3 grain as a function of temperature during the same solidification experiment. |
These experiments provide the first in situ information about the nucleation and growth kinetics of individual grains within the melt during solidification and confirm the mechanism responsible for grain refinement during solidification for the investigated aluminium alloys.
References
[1] J.W. Christian, The theory of transformations in metals and alloys, Pergamon, Oxford (2002).
[2] M. Easton. and D. StJohn, Metall. Mat. Trans. A 30, 1613-1623 (1999).
Principal Publication and Authors
N. Iqbal (a), N.H. van Dijk (a), S.E. Offerman (b), M.P. Moret (c), L. Katgerman (b), G.J. Kearley (a), Acta Mater. 53, 2875-2880 (2005).
(a) FAME, Faculty of Applied Sciences, Delft University of Technology (Netherlands)
(b) Department of Materials Science and Engineering, Delft University of Technology (Netherlands)
(c) ESRF



