- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2005
- Materials Science
- A High-pressure Structure in Curium linked to Magnetism
A High-pressure Structure in Curium linked to Magnetism
The actinide series, elements after actinium and containing Th, U, Np, Pu, Am, Cm, etc., are a fascinating series that are characterised by the progressive filling of the 5f electron shell. Our understanding of these elements is greatly advanced by modern theory that can simulate the complex electron interactions in the solid. However, studies of these materials under extreme conditions, such as pressure, test such theories. As the materials are compressed and the atoms move closer together the interactions increase, resulting in dramatic changes in crystal structure and other properties. A challenge is to determine such structures and compare them with theoretical predictions.
Enormous advances in examining materials at high pressures have been possible because of the development of diamond-anvil technology and the third-generation synchrotron sources, such as the ESRF. However, the application of this technology to radioactive (and toxic) materials is far from straightforward. These experiments have been performed up to 100 GPa at the ID30 beamline on protactinium (Pa), uranium (U), [1] americium (Am) [2] and latterly on curium (Cm).
Figure 36 shows that the compressibility (variation of V/V0 with pressure) at low pressure of Am and Cm, on the one hand, and U and (Pa, not shown but similar to U) on the other hand, are quite different. This is because, for the early actinides Pa to Pu, the 5f states contribute to the “bonding” and they make the atomic volumes small, and the materials “hard”. For the heavier actinides, Am and beyond, the 5f states are “localised” and these materials are subsequently “soft”. However, at a certain pressure the 5f states actually change their character, and we see a “harder” material developing, as in the case of Am [2] at 18 GPa, and Cm above 36 GPa.
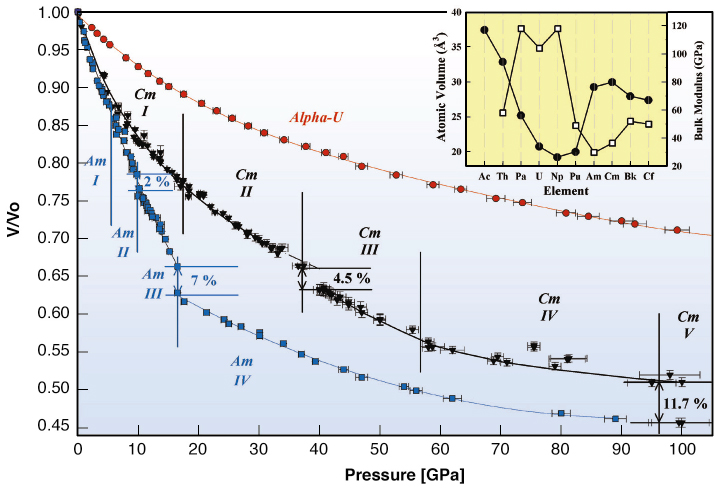 |
|
Fig. 36: Relative volume versus pressure for curves of |
There is no reason that Am and Cm should be different (except that Cm should be slightly less compressible), but it can be seen that Cm has five phases up to 100 GPa, whereas Am has only four. It was quickly established that the first two and last two phases of both elements are identical. The odd phase is Cm-III, which exists between 36 and 56 GPa. The diffraction pattern for this phase is complex; the structure has a low monoclinic symmetry with space group C2/c. This structure has never been found for any element with f electrons. The diffraction pattern, together with the Rietveld fit are shown in Figure 37, with the monoclinic structure of Cm III in the inset.
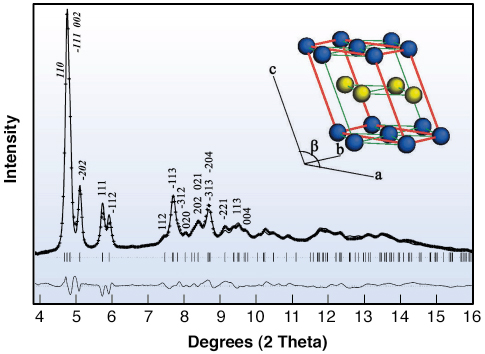 |
|
Fig. 37: Rietveld refinement of Curium III at 45 GPa showing calculated reflection tick marks, principal reflections and difference profile. The insert shows a model of this monoclinic (s.g. C2/c) structure. |
With the structure identified, the question became why this phase should appear in Cm and not in Am? Modern calculations, while unable to predict structures from first principles for heavier elements, are able to compare the ground-state energies from different crystal structures. In these calculations for Cm, an unusual situation emerged. It appears that the Cm-III phase is stable because of the special half-filled nature of the 5f shell, and the associated magnetism present in Cm, and which is not present in Am or any of the preceding actinide elements. Am, for example, has 6 5f electrons that fill the j = 5/2 subshell and result in no ordered magnetism. Normally, magnetic interactions have small energies compared to structural terms, but this is a rare case in which the energy added by the magnetic interaction is sufficient to stabilise a completely new structure.
With the discovery of the Cm-III structure and the prediction that it is stabilised by magnetic interactions, a new perspective is opened on the heavier actinides and by inference (although certainly at even higher pressures) also on the heavy rare-earth metals.
References
[1] T. Le Bihan et al,. Phys. Rev. B 67, 134102 (2003).
[2] S. Heathman et al,. Phys. Rev. Lett, 85, 2961(2000).
Principal Publication and Authors
S. Heathman (a), R.G. Haire (b), T. Le Bihan (c), A. Lindbaum (d), M. Idiri (a), P. Normile (a), S. Li (e), R. Ahuja (e), B. Johansson (e), and G.H. Lander (a), Science 309 110 (2005).
(a) European Commission, JRC, Institute for Transuranium Elements, Karlsruhe (Germany)
(b) Oak Ridge National Laboratory, Tennessee (USA)
(c) CEA-Valduc, DRMN/SEMP/LECP, Is-sur-Tille (France)
(d) Vienna University of Technology, Institute for Solid State Physics (Austria)
(e) Department of Physics, Uppsala University (Sweden)



