- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2005
- X-ray Imaging and Optics
- Evaluation of Gd-EOB-DTPA Uptake in a Perfused and Isolated Mouse Liver Model
Evaluation of Gd-EOB-DTPA Uptake in a Perfused and Isolated Mouse Liver Model
Gd-EOB-DTPA is a paramagnetic complex designed as a hepatotropic magnetic resonance imaging (MRI) contrast agent (CA) useful to evaluate liver functions and pathologies [1]. Even if contrast-enhanced MRI is recognised as a powerful tool for the assessment of functional parameters, the quantitative interpretation of the CA effect on image intensity is not straightforward because the MR signal is a complex function of CA characteristics and image acquisition parameters. Depending on its concentration and imaging sequences, a given CA can indeed induce an opposite effect on contrast. For these reasons, CA concentration is difficult to obtain from image intensity measurements. Among computed X-ray tomography methods, monochromatic quantitative computed tomography (MQCT) has been implemented at ESRF (medical beamline, ID17) and highlighted as a complementary tool to other imaging techniques in the field of preclinical research [2]. As a result of the monochromaticity of the synchrotron beam, this technique allows for nondestructive, real-time, and absolute quantitative in vivo measurement of Gd concentration. The aim of this study was to evaluate the pharmacokinetic pattern of Gd-EOB-DTPA in a model of isolated and perfused mouse livers by both MRI and MQCT techniques.
Hepatic perfusion consists of the administration to the liver of a blood substituting solution. Three protocols of mice liver perfusion were carried out for MRI experiments (gallbladder spared, severed or clamped) whereas the gallbladder was always spared for MQCT experiments. Gd-EOB-DTPA solution was directly administered through the perfusion medium as follows: for MQCT imaging at a final concentration of 0.26 and 0.52 mM of perfusion medium, and for MRI imaging at the optimal case obtained in MQCT, i.e., 0.52 mM. Mice livers were perfused during 1 hour, with the first 30 minutes in recirculating mode (accumulation of CA) and the following 30 minutes in open circuit (washout period). MRI and MQCT images were acquired during the one-hour perfusion period.
 |
|
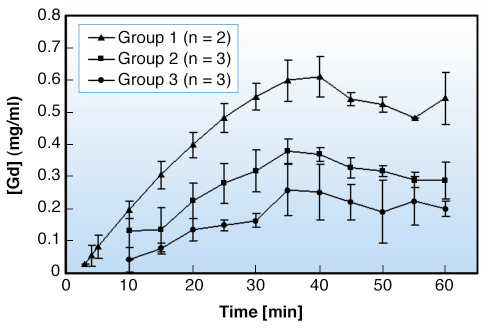
Fig. 147: Pharmacokinetic curves obtained by MQCT. |
Figure 147 presents the quantitative results obtained in MQCT after image analysis. At the highest dose, two groups of mice are distinguishable according to the accumulation kinetics of Gd-EOB-DTPA (represented by ![]() and
and ![]() ). As expected, the curve obtained at the lowest dose exhibits the smallest evolution (represented by
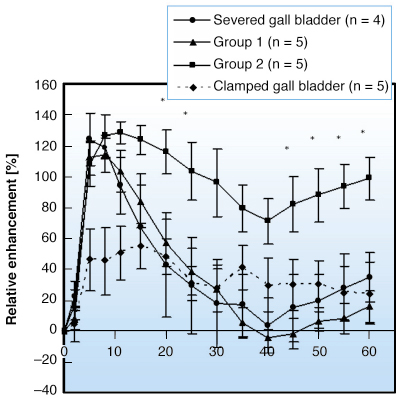
). As expected, the curve obtained at the lowest dose exhibits the smallest evolution (represented by ![]() ). At both doses, the kinetic curve of Gd-EOB-DTPA shows the same characteristic: a phase of uptake (increase of liver intensity that reaches a plateau) followed by the clearance of the CA (decrease of the intensity). As in MQCT, two clear trends were also observed in MRI with regard to the accumulation and elimination of Gd-EOB-DTPA when the gallbladder was spared (Figure 148). The first group exhibited a larger contrast evolution than the second one, in which signal intensities decreased later and less rapidly. Although quantitatively different, these two behaviours have a similar signature characterised by three distinct phases. The first phase is a significant contrast uptake. The increase of signal intensity is caused by a T1 effect resulting from the accumulation of the CA. The subsequent phase (from the 8th to the 40th minute of perfusion) is characterised by a decrease of intensity, slower in the second group than in the first one. This decrease of intensity is caused by the well-known T2 effect resulting from a too high concentration of CA accumulated by the liver. Finally, the clearance of the contrast agent is characterised by a restoring of the signal intensity; this is the return to a T1 effect resulting from the progressive elimination of Gd-EOB-DTPA. Two complementary experiments were performed with MRI to understand the behaviours described here (Figure 148). Livers with severed gallbladder follow the same behaviour as livers of the first group while contrast agent accumulation is impaired in livers with clamped gallbladder (low increase of signal intensity).
). At both doses, the kinetic curve of Gd-EOB-DTPA shows the same characteristic: a phase of uptake (increase of liver intensity that reaches a plateau) followed by the clearance of the CA (decrease of the intensity). As in MQCT, two clear trends were also observed in MRI with regard to the accumulation and elimination of Gd-EOB-DTPA when the gallbladder was spared (Figure 148). The first group exhibited a larger contrast evolution than the second one, in which signal intensities decreased later and less rapidly. Although quantitatively different, these two behaviours have a similar signature characterised by three distinct phases. The first phase is a significant contrast uptake. The increase of signal intensity is caused by a T1 effect resulting from the accumulation of the CA. The subsequent phase (from the 8th to the 40th minute of perfusion) is characterised by a decrease of intensity, slower in the second group than in the first one. This decrease of intensity is caused by the well-known T2 effect resulting from a too high concentration of CA accumulated by the liver. Finally, the clearance of the contrast agent is characterised by a restoring of the signal intensity; this is the return to a T1 effect resulting from the progressive elimination of Gd-EOB-DTPA. Two complementary experiments were performed with MRI to understand the behaviours described here (Figure 148). Livers with severed gallbladder follow the same behaviour as livers of the first group while contrast agent accumulation is impaired in livers with clamped gallbladder (low increase of signal intensity).
 |
|
Fig. 148: Relative enhancement expressed in percentage (RE%) as a function of time obtained on MRI images. |
In conclusion, this pilot study has confirmed the validity of both approaches for evaluating the uptake of a hepatotropic contrast agent by the mouse liver. MRI is an ideal technique for diagnosis, but the complex relationship between the CA concentration and signal intensity makes it difficult to quantitatively follow pharmacokinetic patterns. Although less conveniently accessible, MQCT is the most direct approach to the quantitative measurement of the concentration of the CA. This study has additionally demonstrated the peculiarity of the model of the isolated and perfused mouse liver, which appears unstable, leading to an abnormal behaviour characterised by an impaired uptake function.
References
[1] H.J. Weinmann, W. Ebert, B. Misselwitz and H. Schmitt-Willich, Eur. J. Radiol. 46, 33-44 (2003).
[2] G. Le Duc, S. Corde, H. Elleaume, F. Esteve, A.M. Charvet, T. Brochard, S. Fiedler, A. Collomb and J.F. Le Bas, Eur. Radiol. 9, 1487-1492 (2000).
Principal Publication and Authors
J. Segers (a), G. Le Duc (b), C. Laumonier (a), I. Troprès (c), L. Vander Elst (a) and R.N. Muller (a), Invest. Radiol. 40, 574-582 (2005).
(a) University of Mons-Hainaut, Mons (Belgium)
(b) Biomedical beamline, ESRF
(c) 3T MRI unit, CHU, Grenoble (France)



