- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2005
- High Resolution and Resonance Scattering
- Oxidation State of Vanadium in Natural V-bearing Titanomagnetite
Oxidation State of Vanadium in Natural V-bearing Titanomagnetite
Vanadium is a widely spread element in nature, which may occur in three different redox states, from trivalent to pentavalent, in terrestrial magmatic systems. Its abundance in rock forming minerals is a valuable geochemical indicator, holding information on the oxygen fugacity (i.e. oxidative) conditions prevailing during the formation of igneous rocks. Vanadium is also an element of significant industrial importance, being a component of high-performance steels (Figure 28). However, ore minerals with vanadium as the primary metal rarely occur in nature. Rather, vanadium is extracted from V-rich titanomagnetite (Fe3–xTixO4) cumulates in the upper part of layered basic intrusions. The largest vanadium ore deposits in the world are found in the Bushveld Complex (Republic of South Africa), which hosts more than one-third of the world’s vanadium reserves. In this giant (450 x 370 x 7 km3) layered intrusion, about 2 billion years old, the metal is encountered in 2 to 10 m thick layers of massive titanomagnetite. However, the chemical form of the vanadium (i.e. oxidation state) in these magnetites is largely unconstrained. Its accumulation in only a few magnetite layers, among 25 identified in the upper part (2 km thick) of the complex, together with the mechanisms leading to the formation of metre-thick monomineralic magnetite layers, are still poorly understood. Experimental vanadium partitioning data between magnetite and iron-bearing silicate liquids suggest that V-rich magnetites may result from the partial crystallisation of a basaltic melt in a narrow range of oxygen fugacity [1].
 |
|
Fig. 28: High-temperature extraction of vanadium from vanadiferous titanomagnetite in South Africa. Insert: Structure of titanomagnetite. Ti occurs in the octahedral sites whereas Fe occupies both tetrahedral and octahedral sites of the spinel-type structure. |
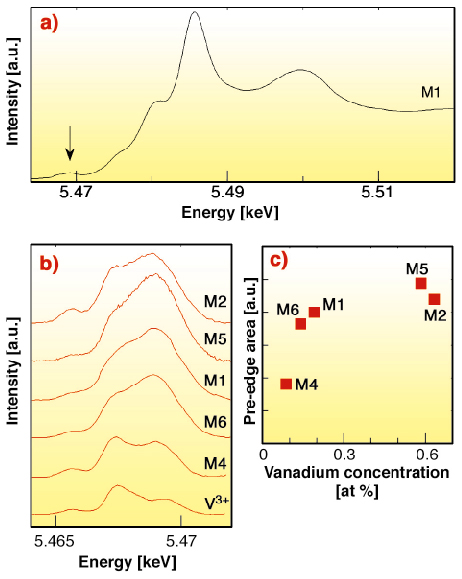
To understand the factors controlling the incorporation of vanadium in magnetite, we have investigated the crystal-chemistry of vanadium in natural titanomagnetite samples from two magnetite seams of the Bushveld complex using X-ray absorption near-edge structures (XANES) (Figure 29). We focused on the pre-edge features observed in the X-ray absorption spectra at the V K-edge, which depend on the valence state and coordination number of transition elements. However, Ti K-fluorescence makes it difficult to obtain high quality XANES spectra in titanomagnetite with a conventional fluorescence setup. The use of the high energy-resolution emission spectrometer installed on the ID26 beamline, based on a spherically bent Ge (331) crystal analyser, made it possible to resolve this difficulty by selecting the V K![]() 1 fluorescence line, hence obtaining the first high energy-resolution XANES spectra of V-bearing titanomagnetite samples at the V K-edge. The resulting sharper pre-edge features [2] show that vanadium is mostly in the trivalent state, by comparison with crystalline references. However, the modification of the pre-edge shape and intensity in the more concentrated samples suggests that they contain a small but significant (~ 10 %) fraction of V4+ (Figure 29). Low magnetite/melt partition coefficients show a preferential trapping of V4+ in the structure of the magmatic liquid. The measured fraction of V4+ thus indicates relatively oxidising conditions during the crystallisation of V-rich titanomagnetite. The occurrence of small quantities of V4+ in the vanadium-rich titanomagnetites of the Bushveld Complex is consistent with previous petrological modelling [1], which shows that extreme enrichments of vanadium in magnetite will only occur for a relatively small range of oxygen fugacity conditions, close to the Ni-NiO buffer. Valence-selective XANES spectroscopy of vanadium in magnetite may thus provide unique information on the redox conditions prevailing during the formation of large-scale magmatic vanadium ore deposits.
1 fluorescence line, hence obtaining the first high energy-resolution XANES spectra of V-bearing titanomagnetite samples at the V K-edge. The resulting sharper pre-edge features [2] show that vanadium is mostly in the trivalent state, by comparison with crystalline references. However, the modification of the pre-edge shape and intensity in the more concentrated samples suggests that they contain a small but significant (~ 10 %) fraction of V4+ (Figure 29). Low magnetite/melt partition coefficients show a preferential trapping of V4+ in the structure of the magmatic liquid. The measured fraction of V4+ thus indicates relatively oxidising conditions during the crystallisation of V-rich titanomagnetite. The occurrence of small quantities of V4+ in the vanadium-rich titanomagnetites of the Bushveld Complex is consistent with previous petrological modelling [1], which shows that extreme enrichments of vanadium in magnetite will only occur for a relatively small range of oxygen fugacity conditions, close to the Ni-NiO buffer. Valence-selective XANES spectroscopy of vanadium in magnetite may thus provide unique information on the redox conditions prevailing during the formation of large-scale magmatic vanadium ore deposits.
 |
|
Fig. 29: (a) V K-edge XANES spectra of a titanomagnetite sample (Bierkraal borehole). Arrow: position of pre-edge features. (b) Pre-edge peaks of titanomagnetite samples after baseline subtraction. For comparison, the pre-edge of a tsavorite crystal, a reference V3+-bearing garnet, is reported at the bottom. The V concentration of titanomagnetite increases from bottom to top. (c) Pre-edge area as a function of vanadium concentration in the Bushveld samples. |
References
[1] M. Toplis, A. Corgne, Contrib. Mineral. Petrol., 144, 22-37 (2002).
[2] P. Glatzel, U. Bergmann, Coord. Chem. Rev., 249, 65-95 (2005).
Principal Publication and Authors
E. Balan (a,b), J.P.R. De Villiers (c), S.-G. Eeckhout (d), P. Glatzel (d), M. Toplis (e), E. Fritsch (a,b), T. Allard (b), L. Galoisy (b), and G. Calas (b), Amer. Mineral., submitted.
(a) UR T058, Institut de Recherche pour le Développement (IRD), Paris (France)
(b) Institut de Minéralogie et Physique des Milieux Condensés (IMPMC),UMR CNRS 7590, Universités Paris VI et VII, IPGP (France)
(c) Department of Materials Science and Metallurgical Engineering, University of Pretoria, (Republic of South Africa)
(d) ESRF
(e) Laboratoire Dynamique Terrestre et Planétaire, UMR CNRS 5562, Observatoire Midi-Pyrénées, Toulouse (France)



