- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- Materials Science
- Design of Crystallised Hybrid Solids with Giant Cells and Mesopores
Design of Crystallised Hybrid Solids with Giant Cells and Mesopores
Porous solids are strategic materials owing to their applications in petroleum chemistry, fine chemicals, catalysis, separation and storage of gases. They exist either as purely inorganic or hybrid organic-inorganic frameworks. The current challenge [1] is to obtain giant pores to use them also as nanoreactors.
For such a challenge, chance is not sufficient, and design procedures must be introduced [2]. This requires the combination of targeted chemistry and computer simulations. This is particularly possible with hybrid solids, which are constructed from inorganic building units and organic moieties. For design purposes, we need first to establish the synthetic chemical conditions corresponding to the existence of targeted inorganic units in solution. Second, we use computer simulations to explore in 3D space the possibilities of a connection between the above inorganic unit with the organic ligand (here 1,3,5-benzene tricarboxylate [BTC]). The simulations yield candidate crystal structures with their atomic coordinates and space groups. Then a comparison between the X-ray powder diffraction patterns of the different simulated models and the experimental one allows us to identify the correct candidate.
This new procedure was applied to µ3-O chromium(III) octahedra trimeric building units with BTC. The resulting product (MIL-100) was a green powder with a Langmuir surface of ~ 3,100 m2/g indicating its porous character. The chemical analysis of this product gave a ratio of 1:2 between Cr trimers and BTC. This information was used in the simulation program. The latter provided three candidate polymorphs with close lattice energies, together with their theoretical X-ray powder diffraction patterns. The position of the Bragg peaks of only one of them fit (Figure 24) those of the experimental product (labelled MIL-100). It consists of a cubic unit-cell (S.G. Fd-3m, a = 72.26 Å, V = 387,000 Å3) far beyond the current possibilities for solving the structure from ab initio procedures.
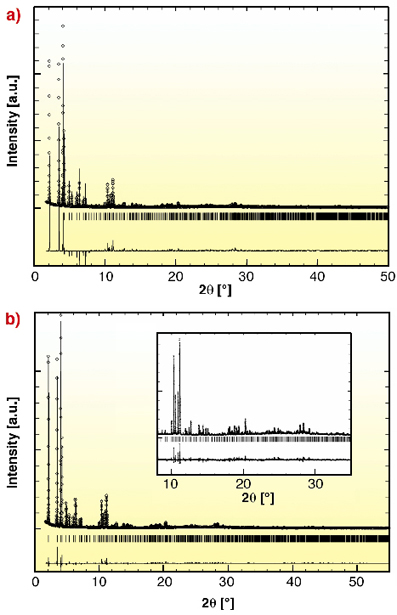 |
Fig. 24: Comparison of the theoretical (a) and refined experimental (b) patterns of MIL-100. |
Starting from the coordinates provided by the simulations, the Rietveld refinement of the crystal structure (68 independent framework atoms) was performed from synchrotron data collected at 80 K on beamline ID31 (![]() : 1.55075 Å) and further allowed us to localize the extra-framework water molecules with good R values. The difference in intensities relates mainly to the fact that the simulation takes into account only the framework and not the species inserted in the cages.
: 1.55075 Å) and further allowed us to localize the extra-framework water molecules with good R values. The difference in intensities relates mainly to the fact that the simulation takes into account only the framework and not the species inserted in the cages.
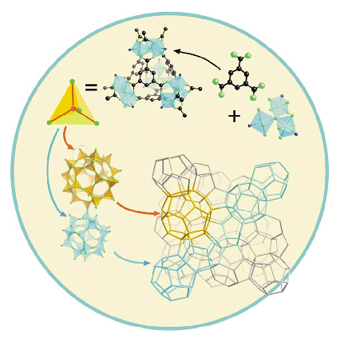 |
Fig. 25: Genesis of the structure from Cr(III) trimers and BTC forming first supertetrahedra which connect in three dimensions to give cages with 20- and 28 supertetrahedra. |
Beside the originality of the method, the structure of MIL-100 itself is appealing. Indeed, the trimers and the BTC molecules (Figure 25) are first linked to each other via the carboxylate functions in order to build supertetrahedra whose corners are occupied by the trimers and the faces by BTC. These supertetrahedra are microporous with free diameters of ca. 6.6 Å. More interestingly, they connect to each other via the trimers in order to build a three-dimensional structure of corner-sharing supertetrahedra with two types of cages within the framework whose dimensions are now typically in the range of mesopores. The smallest, limited by 20 supertetrahedra and forming pentagon dodecahedra, has an internal free diameter of ~ 25 Å and pentagonal windows: (~ 4.8 x 5.8 Å). The face sharing of these dodecahedra creates larger cavities, limited this time by 28 supertetrahedra. The polyhedron determined by the centres of the supertetrahedra has now 12 pentagonal and 4 hexagonal faces. The free aperture of the large hexagonal windows is ~ 8.6 x 8.6 Å and the internal free diameter close to ~ 29 Å. In the as-prepared materials, the cavities are occupied only by water molecules, which evolve from room temperature to 100°C.
In conclusion, MIL-100 represents the largest non proteinic solid isolated to date, the structure of which could be solved only by applying an original method combining targeted chemistry, computer simulation and powder diffraction. Using this approach, the resolution of structures, even very complex ones, is restricted to a Rietveld refinement. Moreover, MIL-100 provides the first example of a mesoporous solid with crystalline walls, at variance to the usual mesoporous solids, with a unique hierarchical system of three types of cages of different dimensions. Also, MIL-100 may have outstanding applications in the domain of sorption/separation of species.
References
[1] G. Férey, A.K. Cheetham, Science 283, 1125 (1999).
[2] M. Eddaoudi, D.B. Moler, H.Li, B. Chen, T.M. Reineke, M. O'Keeffe, O.M. Yaghi, Acc. Chem. Res. 34, 319 (2001)
Principal Publication and Authors
G. Férey (a), C. Serre (a), C. Mellot-Draznieks (a), F. Millange (a), S. Surblé (a), J. Dutour (a), I. Margiolaki (b), Angew. Chem. Int. Ed. 43, 6285, 6290, 6296 (2004) (3 articles).
(a) Institut Lavoisier, UVSQ, Versailles (France)
(b) ESRF



