- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- Materials Science
- Single-bonded Cubic Form of Nitrogen
Single-bonded Cubic Form of Nitrogen
Nitrogen usually consists of molecules where two atoms are strongly triple-bonded. Here, we report on a new form of nitrogen where all atoms are connected with single covalent bonds, similar to carbon atoms in diamond. The compound was synthesised directly from molecular nitrogen at temperatures above 2,000 K and pressures above 110 GPa using a laser-heated diamond cell. From X-ray and Raman scattering we have identified this as the long-sought-after polymeric nitrogen with the theoretically predicted cubic gauche structure (cg-N). This cubic phase has not been observed previously in any element. The phase is a hard substance with bulk modulus ~ 300 GPa, characteristic of strong covalent solids. The cg-N represents a new class of single-bonded nitrogen materials with unique properties such as energy capacity: more than five times that of the most powerfully energetic materials.
It was predicted that at high pressure, solid molecular nitrogen would transform to an atomic solid with a single-bonded crystalline structure called polymeric nitrogen [1,2]. Experimentally, a new, dark, apparently non-molecular phase has been recently found above 180 GPa at 80 K [3] and then at room and elevated temperatures [4,5]. Some properties of this black phase are close to that predicted for the polymeric nitrogen; that is, the value equilibrium pressure (about 100 GPa), and a huge hysteresis enabling the black phase to be recovered at ambient pressure and low temperatures. However, there are no convincing data for cg-N or any other phase with an extending polymeric network.
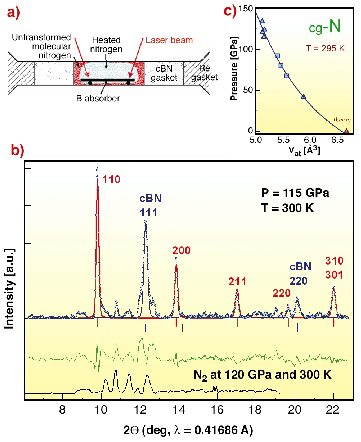 |
|
Fig. 43: X-ray diffraction measurements of nitrogen after heating to 2,600 K at 140 GPa. (a) A cross-section of the sample arrangement. X-ray diffraction was measured with a monochromatic X-ray beam with 0.4168 Å wavelength focused to a spot of ~5 x 5 µm. Diffraction spectra were taken across the culet in 5 µm steps. The spectrum shown in (b) was from the centre of the sample which was oscillated at ±1° around the vertical axis to average reflections. Positions and intensities of the peaks were fitted using Rietveld refinement for the cg-N phase (red line) and c-BN (blue solid line). The green spectrum is the difference between the refinement and the experimental spectrum. This molecular phase was not refined because its structure was not known. For comparison, a spectrum of another, unheated sample at 120 GPa is shown at the bottom. (c) The EOS of the cubic gauche structure (cg-N). The experimental points were fitted with BM EOS with B42 = 460.72 GPa, B'42 = 4.0, V42 = 5.878 Å3 taken at 42 GPa. Extrapolation of this EOS to zero pressure gives a volume of cg-N structure about 6.6 Å3 - in excellent agreement with theoretical predictions of 6.67 Å3 [2]. The zero-pressure bulk modulus B0 calculated using different forms of EOS was found to lie in the range 300 340 GPa. |
We synthesised polymeric nitrogen in numerous runs at T > 2,000 K at P > 110 GPa. The cell was specifically designed for X-ray diffraction measurements of such a weak scatterer as nitrogen (Figure 43a). We used c-BN gasket which produces only a few weak peaks, and the 1-µm thick boron plate (a very weak scatterer) as an absorber of the laser radiation. The highly-focussed X-ray beam of bealine ID09 was used to scan the sample. We fitted the measured spectra to a mixture of cg-N and c-BN (Figure 43b). Parameters from Rietveld refinement for cg-N were: space group I 213, a0 = 3.4542 Å, sites 8a, x = 0.067. The cg-N structure is shown schematically in Figure 44. All nitrogen atoms are three-fold coordinated and bond-lengths are the same for all pairs of bonded atoms. At a pressure of 115 GPa, the bond length is 1.346 Å. We also determined the cg-N structure on decreasing pressure and found its bulk modulus B0 ~ 300 GPa.
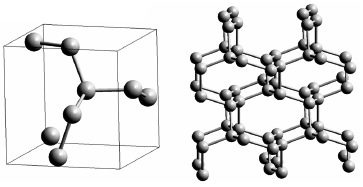 |
Fig. 44: The cg-N structure. Each atom of nitrogen is connected to three neighbours with three single covalent bonds. The primitive cell is shown on the left, and an extended structure of the polymeric nitrogen is shown on the right. |
References
[1] A. K McMahan, and R. LeSar Phys. Rev. Lett. 54, 1929 (1985).
[2] C. Mailhiot, et al Phys. Rev. B 46, 14419 (1992).
[3] M. Eremets. et al Nature 411, 170 (2001).
[4] A.F. Goncharov et al Phys. Rev. Lett. 85, 12621265 (2000);
[5] E. Gregoryanz et al Phys. Rev. B 64, 052103 (2001), ibid B 66, 224108 (2002).
Principal Publication and Authors
M.I. Eremets (a), A.G. Gavriliuk (abc), I.A. Trojan (ac), D.A. Dzivenko (a), R. Boehler (a), Nature Materials 3, 558 (2004).
(a) Max Planck Institute für Chemie, Mainz (Germany)
(b) A.V.Shubnikov Institute of Crystallography of Russian Academy of Sciences, Moscow (Russia)
(c) High Pressure Institute of Russian Academy of Sciences, Troitsk (Russia)



