- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- X-ray Imaging
- Modern Composition of 3,500 Million-year-old Seawater: Implications for Life on the Primitive Earth
Modern Composition of 3,500 Million-year-old Seawater: Implications for Life on the Primitive Earth
There are two fundamental requirements for life as we know it, liquid water and organic polymers, such as nucleic acids and proteins. At about 4.4 to 4.0 billion years (Gyr), the Earth may have been cool enough for the presence of extensive oceans for long periods. Life on Earth dates from before about 3.8 Gyr, and is likely to have gone through one or more hot-ocean environments. Only hyperthermophiles (microorganisms optimally living in water at 80110°C) would have survived. Early hyperthermophile life probably developed near hydrothermal systems including 'black smokers' (Figure 135a). It would have had access to redox contrast between a more oxidised atmosphereocean system and the more reduced fluids emanating from mantle-derived magmas. Sulphur offers the best opportunities. With water present in the air, volcanic SO2 would have been oxidised to sulphate. Dissolved in water, sulphate would provide oxidation power for organisms to react against reduced species in hydrothermal fluids, such as H2S and CH4. Considering that sulphate-reducing microbes had evolved by 3.5 Gyr ago, the poorly-fractionated Early Archaean sedimentary sulphides have been attributed to either biological sulphate reduction in a fully anoxic ocean containing little sulphate (< 200 µM to 1 mM) or to intense biological sulphate reduction at sulphate oceanic concentrations not much different from today's (28 mM). Clearly, the oxidation state and composition of the Archaean ocean and atmosphere remains essentially unconstrained.
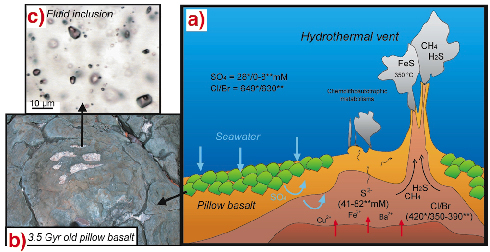 |
|
Fig. 135: (a) Hydrothermal system on the seafloor showing the main fluid pathways and elemental distribution patterns. * refers to modern day values, whereas ** corresponds to element concentration estimates in "North Pole" Archaean fluids. (b) Archaean pillow basalt. (c) Fluid inclusions in intrapillow quartz pods. |
Insights into the composition of Archaean seawater and hydrothermal fluids can be obtained by direct analysis of fluid inclusions preserved in Archaean hydrothermal systems. Here, we investigated a suite of quartz pods preserved in basaltic pillow lavas (Figure 135b) that formed during oceanic hydrothermal alteration of the 3.5 Gyr North Pole formation. The pods contain myriad fluid inclusions (Figure 135c), which were analysed individually using synchrotron radiation X-ray microfluorescence (µ-SR-XRF) [1]. The main advantages of the µ-SR-XRF technique reside in its non-destructive character, high spatial resolution, multi-element analytical capability and high sensitivity. Hence, µ-SR-XRF can analyse small, diluted individual fluid inclusions, discriminate distinct inclusion populations trapped in a single crystal and provide information about complex histories of fluid circulations [2].
Calculated model composition yielded a bulk fluid salinity of four-times the present-day value reflecting a typical modern-day seawater evaporation trend in a shallow marine, closed basin environment. Individual fluid inclusion analysis using µ-SR-XRF revealed the presence of three fluid populations (Figure 136): a metal-depleted fluid, a Ba-rich and S-depleted fluid, and a FeS-rich end-member. The Cl/Br ratio of metal-depleted fluid inclusions (630) is similar to the modern seawater value (649; Figure 135a). In contrast, Ba- and Fe-rich brines have Cl/Br ratios (350 and 390) close to the bulk Earth value (420), hence arguing for a hydrothermal origin of these fluids. The metal depleted fluid displays low sulphate concentration (08 mM compared to 28 mM in present-day ocean). Sulphur content of the Fe rich fluids ranges between 41 and 82 mM.
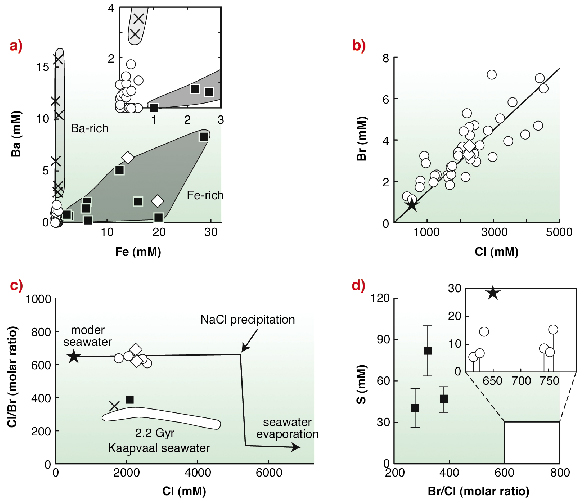 |
|
Fig. 136: Fluid inclusion composition results. (a) Fe vs. Ba concentration diagram showing the occurrence a Ba-rich fluid (crosses), a Fe-rich fluid (black squares) and a metal-depleted end-member (open circle) corresponding to "North Pole seawater". (b) Cl vs. Br concentrations for metal-depleted inclusions compared with modern seawater value (black star) and seawater Cl/Br evaporative trend (black line). (c) Cl/Br vs. Cl trend of evaporating seawater compared with present results. Also shown is the field of Kaapvaal 2.2 billion years old seawater, South Africa [3]. (d) S vs. Cl/Br plot of "North Pole seawater" and Fe-bearing inclusion fluids. Black squares = sulphur concentrations in Fe-rich fluids. Open circles = calculated sulphur detection limit and possible range of S concentrations in "North Pole seawater". |
Fluidrock interaction processes occurring in pillow basalts located on the seafloor are unlikely to be the cause of Cl/Br fractionation between hydrothermal fluids and the North Pole seawater end-member. Modern organic matter is strongly enriched in heavy halogens with respect to Cl. If the sedimental burial of organic matter is efficient enough to preclude return of Br to the bottom ocean water, Br can be significantly enriched in sediments and depleted in seawater. We hypothesize that high biological activities associated with vigorous sedimentation are responsible for the buffering of North Pole seawater Cl/Br value. The sulphate concentration (0 to 8 mM, Figure 135), is much lower than in present-day oceans (28 mM) and in the Black Sea anoxic bottom waters (18 mM). Under an oxygen-rich Archaean atmosphere, the relative isolation of the North Pole basin and its exposure to large detrital input, would make it a very favourable setting for the accumulation of sulphate produced by subaerial sulphide oxidation. Therefore the sulphate content of North Pole seawater may be regarded as indicative of local conditions and representing an upper limit for the rest of the Archaean ocean. This implies that the Early Archaean ocean as a whole would have been dramatically depleted in sulphate, hence arguing for a poorly oxygenated primitive atmosphere.
References
[1] J. Cauzid, P. Philippot, A. Somogyi, A. Simionovici, P. Bleuet, Anal. Chem., 76, 3988-3994 (2004).
[2] J. Foriel, P. Philippot, P. Rey, A. Somogyi, D. Banks, B. Ménez, Earth Planet. Sci. Lett., in press (2005).
[3] J. Gutzmer, D.A. Banks, V. Luders, J. Hoefs, N.J. Beukes, K.L. Von Bezing, Chem. Geol., 201, 37-53 (2003).
Authors
P. Philippot (a), J. Cauzid (a, b), J. Foriel (a), A. Somogyi (c), B. Ménez (a), P. Rey (d)
(a) Institut de Physique du Globe de Paris (France)
(b) ESRF
(c) Synchrotron Soleil, Gif-sur-Yvette (France)
(d) School of Geosciences, The University of Sydney (Australia)



