- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- X-ray Imaging
- Synchrotron Photoactivation of Platinum Against Brain Tumours
Synchrotron Photoactivation of Platinum Against Brain Tumours
Despite significant advances in cancer therapy, treatment of high-grade gliomas is still palliative. To date, the median survival for patients with glioblastoma is less than one year. Brain tumour treatment generally includes surgery, radiotherapy and chemotherapy. Although radiotherapy enhances tumour local control, the radiosensitivity of the healthy tissues does not permit the use of a sufficiently high X-ray dose to treat such radioresistant tumours. Furthermore, adjuvant chemotherapy has predominantly failed to improve patient survival mainly because of the difficulty for these drugs to penetrate through the blood-brain barrier.
Among recent therapeutic techniques, photoactivation therapy (PAT) consists in selectively introducing a high-Z atom into the DNA of tumour cell and irradiating with X-rays of suitable energy for an enhanced photoelectric effect on this atom. Thus, the local toxicity is increased. A monochromatic radiation beam would potentially optimise this physical effect. Therefore, synchrotron radiation appears to be the optimal tool for irradiation: its high fluence (105 times brighter than conventional X-ray sources) allows the beam to be tuned to a convenient wavelength while keeping a sufficient flux to allow radiotherapy applications. PAT experiments have been recently performed at beamline ID17. These experiments on F98 rat glioma demonstrated that synchrotron irradiation of a widely-used chemotherapy drug called cisplatin at the platinum absorption K-edge results in an increased toxicity most likely due to an enhanced photoelectric effect on the platinum atoms [1].
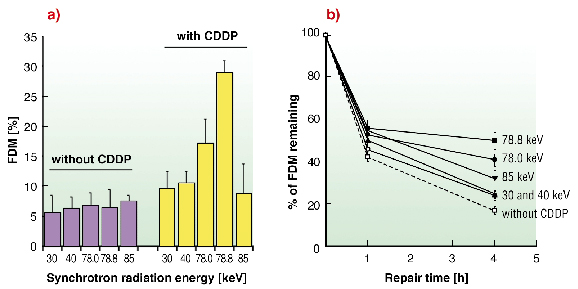
In vitro experiments were performed on F98 cells treated with cisplatin and irradiated at 30, 40, 78, 78.8 or 85 keV. The induction of DNA double strand breaks (DSB) and their subsequent repair were studied in function of the energy of the beam (Figure 150). Cells without cisplatin lead to the same DSB rate whatever the radiation energy tested (Figure 150a). However, when combined with cisplatin, cells irradiated at 78.8 keV (just above the platinum K-edge) showed two and three times more DSB than at 30, 40, 85 and 78 keV, respectively. Furthermore, 4 hours after treatment, the DSB rate was found to be higher at 78.8 keV (Figure 150b). Therefore we demonstrated that cisplatin treatment of F98 cells and irradiation at 78.8 keV, results in a greater amount of more slowly repaired DNA double strand breaks. These conclusions were consistent with those obtained previously in SQ20B human cell line with the same treatment [2].
 |
|
Fig. 150: (a) Yields of DSB induced (expressed in fraction of DNA fragments migrating out of the gel (FDM)) after treatment of F98 cells irradiated (30 Gy) at different energies in presence of cisplatin. (b) DSB repair data as a function of post-irradiation time. |
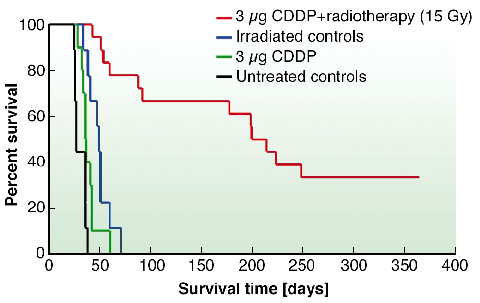
In vivo experiments were thereafter performed on rats inoculated with F98 glioma cells. On day 13 after stereotactic tumour inoculation, 3 µg of cisplatin were injected intra-cerebrally at the tumour site. Rats were irradiated on day 14, with 78.0 and 78.8 keV photons. Median Survival Time (MeST) of untreated rats was 26 days (Figure 151). When cisplatin or synchrotron radiation alone was applied, the MeST was found to be 37 and 48 days, respectively. When both treatments were combined, a very large increase in life span was obtained (MeST = 206.5 days) compared to the controls. One year after treatment, 6 out of the 18 rats treated with CDDP and radiation, were still alive. Furthermore, histological studies performed on these surviving rats did not show any trace of residual tumour suggesting its irreversible regression. This outstanding life span increase (694 %) is the largest obtained to date with the F98 glioma model. Previously, Barth et al. reported an relative increase in life span of 188% using Boron Neutron Capture Therapy [3].
 |
|
Fig. 151: Kaplan-Meier survival curves of rats bearing F98 glioma and subjected to the indicated treatments. |
To conclude, our data demonstrated the efficiency of an innovative anticancer treatment involving both cisplatin and synchrotron radiation. In vivo experiments showed that the combination of cisplatin with synchrotron radiation resulted in the longest reported survival for this radioresistant glioma model. Furthermore, our most recent experiments demonstrate that a synchrotron radiation source is more efficient than a conventional X-ray source used at a hospital for treatment involving cisplatin. This innovative therapeutic method opens new perspectives for brain tumour treatment.
References
[1] M. C. Biston, A. Joubert, J. F. Adam, H. Elleaume, S. Bohic, A. M. Charvet, F. Estève, N. Foray, J. Balosso, Cancer Res, 64, 2317-2323 (2004).
[2] S. Corde, J. Balosso, H. Elleaume, M. Renier, A. Joubert, M. C. Biston, J. F. Adam, A. M. Charvet, T. Brochard, J.F. Le-Bas, F. Estève, N. Foray, Cancer Res, 63, 3221-3227 (2003).
[3] R.F. Barth, W.Yang, J.H. Rotaru, M.L. Moeschberger, C.P. Boesel, A.H. Soloway, D.D. Joel, M.M. Nawrocky, K. Ono, J.H. Goodman, Int J Radiat Oncol Biol Phys, 47, 209-218 (2000).
Author
M.C. Biston on behalf of the INSERM U647 RSRM and ID17 Team Members.
INSERM-RSRM CHU/ESRF, Grenoble (France)



