- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- High Resolution and Resonance Scattering
- Fe K Pre-edges Revealed by Resonant X-ray Emission
Fe K Pre-edges Revealed by Resonant X-ray Emission
The pre-edge structures at the K absorption edge of transition metals obtained from X-ray absorption spectroscopy (XAS) are known to be extremely sensitive to the chemical and structural local environment, and can be used as a fingerprint of the transition metal properties[1]. Compared to L edge measurements, this method is advantageously bulk-sensitive, because it is performed in the hard X-ray energy range, and can be applied to a vast variety of materials such as bio-complexes and Earth science related materials, under various sample environments.
In XAS, however, the pertinence of data analysis hinges on the broadening effect related to the 1s core-hole lifetime. This limitation can be overcome to a certain degree by using a second order process instead, namely resonant X-ray emission spectroscopy (RXES). This method consists of measuring a specific decay channel on resonance as the incident energy is tuned to an absorption edge. As a benchmark of this technique, we have applied RXES in a series of Fe-bearing minerals. We focused on the K![]() emission line (2p
emission line (2p ![]() 1s transition) in the vicinity of the Fe-K edge (1s2p-RXES). In these model systems, Fe can be either of valence 2+ or 3+ and sits in a supposedly pure octahedral (Oh) or tetrahedral site (Td). All the spectra were obtained on the ID26 beamline using a Rowland circle spectrometer and a spherically-bent Si analyser.
1s transition) in the vicinity of the Fe-K edge (1s2p-RXES). In these model systems, Fe can be either of valence 2+ or 3+ and sits in a supposedly pure octahedral (Oh) or tetrahedral site (Td). All the spectra were obtained on the ID26 beamline using a Rowland circle spectrometer and a spherically-bent Si analyser.
Figure 20 shows the absorption edges obtained by RXES in the partial fluorescence yield mode (PFY). Here, the emitted energy E2 is kept fixed at the maximum of the K![]() line while the incident energy E1 is varied through the Fe-K absorption edge. The presence of a shallower core-hole in the RXES final state, when compared to XAS, yields to a remarkable sharpening effect. Notably, the pre-edge exhibits a clear dependence on both Fe site-symmetry and valence. In a simple mono-electronic approach, this can be understood as due to the lift of degeneracy of the 3d states on the metal site, split by the crystal field of the surrounding ions. In octahedral environment, this would result in a mere doublet state, as observed in andradite. The absence of centre of symmetry in Td ions leads to a more complicated picture: dipolar transitions to d-states hybridised with delocalised p electrons are now allowed. This yields an increase of intensity of the pre-edge by a factor of ~5.
line while the incident energy E1 is varied through the Fe-K absorption edge. The presence of a shallower core-hole in the RXES final state, when compared to XAS, yields to a remarkable sharpening effect. Notably, the pre-edge exhibits a clear dependence on both Fe site-symmetry and valence. In a simple mono-electronic approach, this can be understood as due to the lift of degeneracy of the 3d states on the metal site, split by the crystal field of the surrounding ions. In octahedral environment, this would result in a mere doublet state, as observed in andradite. The absence of centre of symmetry in Td ions leads to a more complicated picture: dipolar transitions to d-states hybridised with delocalised p electrons are now allowed. This yields an increase of intensity of the pre-edge by a factor of ~5.
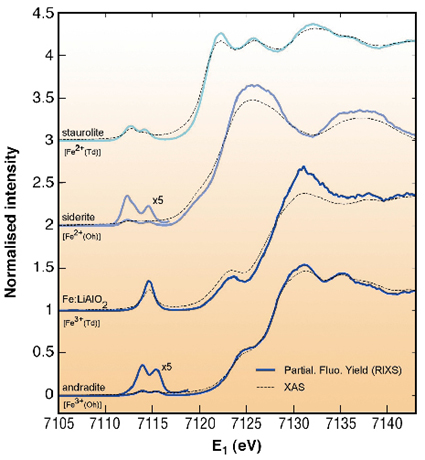 |
Fig. 20: High resolution absorption spectra at the Fe-K edge obtained by RXES in the PFY mode. The PFY-spectra are compared to conventional XAS (dotted lines). |
Better insight can be gained by acquiring a complete map of the pre-edge region, in the RXES sense, as illustrated in Figure 21 (see also [2]) for the four selected models. The colour-interpolated 2D-maps are reconstructed from series of RXES spectra, measured on a fine incident energy grid in the pre-edge region, and shown here as a function of the transfer energy E1-E2. In this plane, PFY absorption spectra correspond to cuts at 45°, while off-diagonal intensity reveals the spread of the 3d multiplet in the final state (in presence of a 2p core-hole), comparable to the Fe L absorption edges. Finally, XAS spectra can be recovered by integration over all transfer energies. RXES maps therefore provide a graphic and extensive representation of the Fe K pre-edge. In andradite, the distance between the two resonances (~1.6 eV) yields a direct estimate of the crystal field parameter (10Dq) in Oh symmetry. Similarly, in Fe:LiAlO2 (Td-Fe3+), the asymmetric tail elongated towards to bottom left is reminiscent of the splitting of the d orbitals in Td symmetry. Note that this 0.7 eV splitting would be entirely masked by lifetime broadening in XAS. Analysis of the pre-edge in Fe2+ compounds implies taking into account multiplet effects, as shown by strong off-diagonal resonances, e.g. in staurolite (Td-Fe2+).
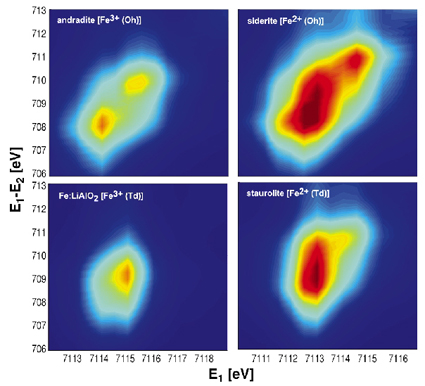 |
Fig. 21: RXES 2D-maps of the pre-edge region in the incident (E1) vs transfer energy (E1-E2) plane for the four minerals. |
In conclusion, the RXES method should be of particular interest for the field of mineralogy. It also merits broader interest for studying the electronic properties of complex materials in constrained environments.
References
[1] M. Wilke et al., Am. Mineral. 86, 714 (2001).
[2] P. Glatzel et al., J. Am. Chem. Soc. 126, 9946 (2004).
Principal Publication and Authors
J.-P. Rueff (a), L. Journel (a), P.-E. Petit (b), F. Farges (c), Phys. Rev. B, 69, 235107 (2004).
(a) Laboratoire de Chimie Physique - Matière et Rayonnement, CNRS - Université Paris 6 (France)
(b) Institut des Matériaux J. Rouxel, CNRS - Université de Nantes (France)
(c) Laboratoire des Géomatériaux, CNRS - Université de Marne la Vallée (France), and Department of Geological & Environmental Sciences, Stanford University (USA)



