- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2003
- X-ray Absorption and Magnetic Scattering
- High-pressure Evolution of the Electronic and Local Structure of Bromine
High-pressure Evolution of the Electronic and Local Structure of Bromine
The diatomic molecule can be considered as the most simple condensed-matter object. In spite of this simplicity, it still reserves many hidden surprises [1]. When diatomic molecules are brought together they can form perfectly arranged structures called molecular crystals. The main characteristic of this class of crystals is that the molecule is preserved and the intra-molecular distance is much smaller than any other interatomic distance within the crystal. Among the molecular crystals, bromine is one of the simplest and is constituted of weakly interacting layers of zig-zag Br2 molecules. The other halogens (I2 and Cl2) share the same crystal structure. What happens when we try to compress this crystal? The best known part of the play is the last act: at a given point the inter-molecular distance becomes comparable to the dimer inter-atomic distance and the molecule is said to dissociate. In general this takes place abruptly through a first-order phase transition and for bromine this happens at the rather high pressure of 80 GPa. All studies on bromine have agreed that the molecule does not show any change up to the dissociation pressure. However, we have recently shown through experiments at the ESRF [2] that the initially semiconducting bromine crystal, metallises at a much lower pressure of about 25 GPa. Is the bromine molecule ignoring these abrupt changes that deeply modify the electronic structure of the crystal? To answer this question we have studied the high-pressure evolution of the bromine molecule with the most sensitive technique for the measurement of short interatomic distances, i.e. X-ray absorption fine structure (EXAFS). The experiments were carried out at ID24, the energy-dispersive EXAFS beamline, where pressure was applied using a diamond-anvil cell. Figure 105 shows the pressure evolution of the EXAFS oscillations that contain the information of the local structure around a bromine atom. The fit of the oscillations was done using a constrained structural model including the crystallographic parameters obtained by high pressure X-ray diffraction experiments and, as shown in Figure 105, the fit is entirely satisfactory. Note that this study constitutes the highest pressure EXAFS experiment ever done. The evolution of the bromine interatomic distance in the molecule is shown in Figure 106. The first surprise is that the evolution of the bromine intra-molecular distance is not monotonic, but it first increases, and then decreases at a higher pressure. We shall recall here that no structural transformation has been observed below 80 GPa. The second surprise is that this change of behaviour takes place precisely at the metallisation pressure. Our results demonstrate that from the electronic point of view we have two totally different types of molecular crystals, above and below 25 GPa, sharing the same crystal structure. We can even be more affirmative and say that after 25 GPa the bromine molecule in a crystal goes a step further in the loss of its molecular character by delocalising part of its electrons in the crystal.
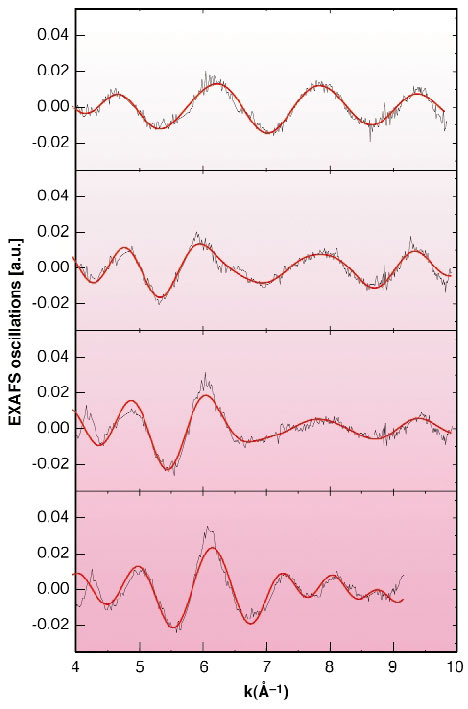 |
|
Fig. 105: EXAFS oscillations of bromine as a function of pressure. |
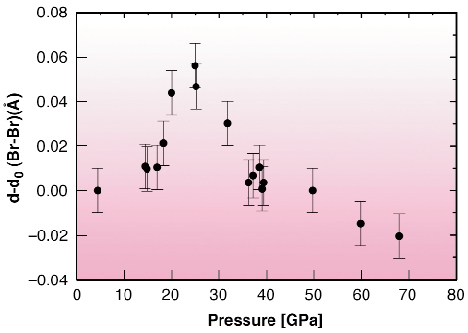 |
|
Fig. 106: Relative evolution of the molecular Br2 distance with pressure. The change of behaviour takes place at the metallisation pressure. |
References
[1] T. Kenichi, S. Kyoko, F. Hiroshi and O. Mitsuko, Nature 423, 971 (2003).
[2] A. San Miguel, H. Libotte, J.-P Gaspard, M. Gauthier, J.-P. Itié and A. Polian, Eur. Phys. J. B 17, 227-233 (2000).
Authors
A. San-Miguel (a), H. Libotte (b), J.P. Gaspard (b), M. Gauthier (c) and S. Pascarelli (d)
(a) Université Lyon 1 and CNRS (France)
(b) Université de Liège, Sart-Tilman (Belgium)
(c) Université Paris VI and CNRS (France)
(d) ESRF



