- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2003
- Materials Science
- Iron-silicon Alloy at Extreme Conditions
Iron-silicon Alloy at Extreme Conditions
The boundary between the Earth's metallic core and its silicate mantle is characterised by sharp changes of density, seismic wave velocities, electrical conductivity, chemical composition, and lateral heterogeneity. Although iron (with ~ 5 wt% Ni) is a dominant component of the Earth's core, Fe-Ni alloy is too dense by ~ 10% for the outer liquid core and by 2-5% for the solid inner core to satisfy the observed density along any reasonable geotherm. On the basis of cosmochemistry, it has been proposed that the core also contains one or more light elements, such as H, C, O, S, and/or Si. It is likely that 2-6 wt% Si was dissolved into the liquid metal during core formation in a magma ocean (pressure around 25 GPa and temperature ~2800 K) during the early history of the Earth. On the basis of a simple thermodynamic model, Gessmann et al. proposed that the solubility of Si at core conditions is close to zero [1]. Thus, following core formation, Si should be expelled from liquid Fe as the metallic core evolves toward chemical equilibrium. Theory predicts that, as pressure increases, the alloy should dissociate to a mixture of silicon-poor hcp Fe and the Si-rich B2 structured phase.
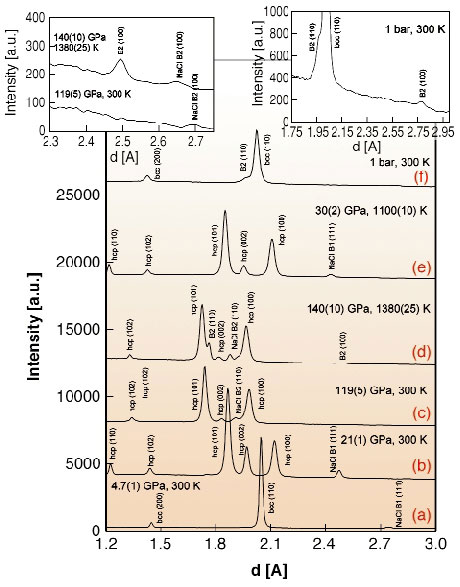
Note that the theoretical calculations were done at T = 0 K, while at high temperature, entropic contribution may be important. Therefore, theoretical predictions were tested through a series of high-P,T DAC experiments on Fe-Si alloys containing 9.6(1), 5.1(1), and 4.3(4) wt% Si. We found that all alloys behave similarly and Figure 53 shows representative X-ray patterns obtained in situ at high-P,T using the 5.1 wt% Si alloy as the starting material. Figure 53d (see also left inset in Figure 53) shows an example of a diffraction pattern collected after 5 hours of external electrical heating of the Fe-5.1 wt% Si alloy at 140(10) GPa and 1380(25) K. Two new lines at 2.494 Å and 1.762 Å belong to the B2 structured alloy (a = 2.493(1) Å) as predicted by ab initio calculations. When the same sample was heated for 8 hours at 30(2) GPa and 1100(10) K, the extra lines disappeared indicating that the reaction was reversed and only the hcp phase remained (Figure 53e).
 |
|
Fig. 53: Examples of X-ray diffraction patterns collected in experiments with Fe 5.1 Si wt%. (a) At 4.7(1) GPa the alloy has bcc structure. Transformation to a hcp phase occurs between 14 GPa and 21(1) GPa (b). No phase transformations were observed on compression to over 119(5) GPa at room temperature (c). However, after five hours of external electrical heating at 140(10) GPa and 1380(25) K (d and left inset), two new lines appeared at 2.494 Å and 1.762 Å belonging to the B2 structured alloy (a = 2.493(1) Å). The same sample was then heated for 8 hours at 30(2) GPa and 1100(10) K (e). The extra lines have disappeared due to back reaction of iron alloys with different concentration and only hcp phase remained. NaCl was used as the pressure medium (reflections marked as NaCl B1 below 30 GPa and NaCl B2 at higher pressures). Spectrum (f) and right inset show an X-ray diffraction patterns of the sample recovered after laser-heating of Fe-10 wt% Si alloy at 105(5) GPa and 2500(100) K. The reflections at 1.969 Å and 2.781 Å correspond to the (110) and (100) peaks of quenched B2 FeSi phase. |
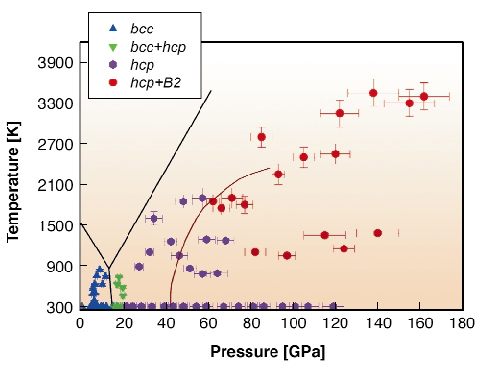
Our results on the behaviour of the Fe-Si alloys in solid and liquid states with different Si concentrations are summarised in Figure 54. We found that at pressures above 60 GPa and high temperatures the alloys dissociate into a mixture of hcp-structured Si-poor and B2-structured Si-rich phases.
 |
|
Fig. 54: Phases observed in electrically- (below 1500 K) and laser- (above 1500 K) heated DAC experiments with iron-silicon alloys (9.6(1), 5.1(1), 4.3(4) wt% Si). We found that all alloys behave similarly and symbols show only phase relations. Continuous black lines show phase relations in pure iron, and a red curve a phase boundary between hcp and hcp+bcc regions in Fe 7.9 Si wt% alloy according to Lin et al. (2002) [2]. |
At the coremantle boundary (CMB) pressure (140 GPa), the density of B2 FeSi (~ 9.0 g/cm3) is significantly lower than the density of the core (~ 10.0 g/cm3) immediately below the CMB and significantly higher than the density of the overlying mantle (~ 5.6 g/cm3). Iron silicide B2 FiSi does not react with silicate perovskite and periclase (or ferripericlase at conditions of Earth's lower mantle. This means that the silicon-rich alloy produced by re-equilibration of proto-core metal that equilibrated with silicates in a magma ocean, or produced at the CMB by reaction between iron and silicate will accumulate at the boundary between the lower mantle and core. Ab initio simulations and measurements on B2 FeSi recovered from multianvil experiments show that this compound is an electric conductor (with measured at ambient conditions conductivity of 6(1) 105 S/m). The presence of B2 FeSi at the base of Earth's mantle could thus explain its anomalously high electrical conductivity and provide a key for understanding why the amplitude of the Earth's nutation is out-of-phase with tidal forcing.
References
[1] C. K. Gessmann, B. J. Wood, D. C. Rubie, M. R. Kilburn, Earth Planet. Science Let. 184, 367-376 (2001).
[2] J.-F. Lin, D. L. Heinz, A. J. Campbell, J. M. Devine, G. Shen, Science 295, 313-315 (2002).
Principal Publication and Authors
L. Dubrovinsky (a), N. Dubrovinskaia (a), F. Langenhorst (a), D. Dobson (a), D. Rubie (a), C. Geßmann (a), I. Abrikosov (b), B. Johansson (b), Nature, 422, 58-61 (2003).
(a) BayerRisches Geoinstitut, Universität Bayreuth (Germany)
(b) Uppsal University (Sweden)



