- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2003
- High Resolution and Resonance Scattering
- Anisotropic Vibrations Labelled by the Haem Iron in Metmyoglobin
Anisotropic Vibrations Labelled by the Haem Iron in Metmyoglobin
Proteins are essential for life and are therefore an outstanding object of research. The function of a protein is determined by its architecture and also strongly by its dynamics. Whereas the structure is nowadays often solved to atomic resolution, the understanding of the dynamics is still inadequate. It is well known, that below a dynamical transition temperature Tc harmonic, solid state like vibrations occur. Above Tc protein specific dynamics appears and superimposes the harmonic vibrations. The nature of this specific dynamics is still under discussion.
In iron containing proteins dynamics can be investigated by many spectroscopic methods. Already thirty years ago Mössbauer spectroscopy on 57Fe was shown to be a valuable tool for the detection of harmonic vibrations and protein specific motions with energies in the neV region in haem proteins [1]. While Mössbauer absorption spectroscopy yields mean square displacements coming from all modes of motions coupling to the iron, Mössbauer effect with synchrotron radiation, also called phonon assisted Mössbauer effect, allows an energy selective measurement of vibrations in the meV energy regime. This suits the investigation of phonons perfectly. Temperature dependent measurements on myoglobin in the deoxy, met and CO-ligated state proved, that the vibrations accessible by this method stay harmonic even above Tc [2]. Recently this method could be extended to investigate anisotropic motions by using a single crystal of myoglobin. Anisotropic mean square displacements of the haem iron as a function of energy are obtained as the projection onto the incoming beam direction.
 |
|
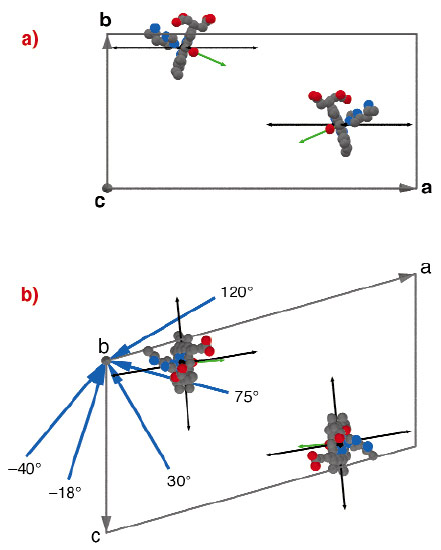
Fig. 16: Sketch of the unit cell of myoglobin. a) View along the c-axes. b) View along the b-axes. Only the haem plane, the ligated oxygen and the His93 are shown with its equivalent in P21 symmetry. Blue arrows around the b-axes indicate the incoming beam direction. The green arrow is the haem normal. The black arrows indicate two selected vibrations enlarged by a factor of 500. |
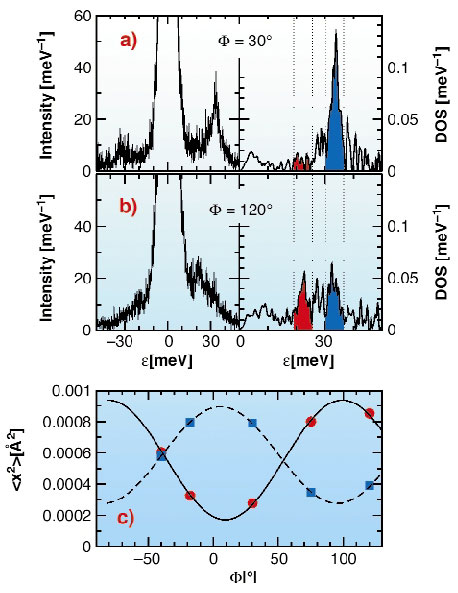
Each unit cell of the metmyoglobin single crystal contains two molecules i.e. about 5000 atoms plus about 800 water. The natural haem iron was enriched in the Mössbauer isotope 57Fe. The protein crystallised in P21 symmetry forming a crystal of 1·2·3 mm3. The crystal was rotated around its crystallographic b-axes perpendicular to the synchrotron beam. Five orientations were measured as sketched in Figure 16. The density of phonon states was extracted from the inelastic scattering spectra, taken at ID18. The mean square amplitude <x2> of modes was calculated within several energy intervals, as exemplary marked in red and blue in Figure 17. A cos2-dependence plus background described the angular dependent projection very well (see Figure 17c). The fit gave the mean vibrational direction of some vibrations as given in Figure 17 exemplary for two vibrations.
 |
|
Fig. 17: a,b) Left: two selected energy spectra at
|
In conclusion, the experiment is a further step in the separation of harmonic vibrations from the protein specific motions by extending the classical Mössbauer absorption to the meV energy regime. Protein-specific motions fall beyond this border and can clearly not be explained by increasing anharmonicity of solid state vibrations. Nevertheless, how the protein-specific motions emerge from the harmonic vibrations with temperature remains unknown. The energy and angular dependence of the vibrations in the active centre will be the basis for a determination of the forces acting in the active centre of a protein.
References
[1] F. Parak, H. Formanek; Acta Cryst. A 27, 573-578 (1971).
[2] K. Achterhold, C. Keppler, A. Ostermann, U. van Bürck, W. Sturhahn, E.E. Alp, F.G. Parak; Phys. Rev. E 65, 051916-1 - 051916-13 (2002).
Principal Publication and Authors
K. Achterhold and F.G. Parak; J. Phys.: Condens. Matter 15, S1683S1692 (2003).
Technische Universität München (Germany)



