- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2003
- High Resolution and Resonance Scattering
- Direct Observation of the Covalency of Liquid Germanium by X-ray Emission Spectroscopy
Direct Observation of the Covalency of Liquid Germanium by X-ray Emission Spectroscopy
The detailed understanding of the electronic structure of liquids remains a challenge, due to the complete lack of long range order. Nevertheless in the last few years a considerable progress in the electronic theory of liquids has been made, mainly based on molecular-dynamic calculations.
On the experimental side, data of the electronic structure of high vapour pressure liquids are still scarce due to severe experimental difficulties. X-ray emission spectroscopy is an alternative method to study the electronic structure of matter under extreme conditions.
Liquid germanium is one of the most often studied liquid metal systems. The element shrinks upon melting and undergoes a semiconductor-metal transition. In contrast to the structure of simple liquid metals with a coordination number around 12, liquid germanium has a lower coordination number around 6 [1]. Among the theoretical models explaining the complex structure of liquid germanium, the one claiming the persistence of covalent bonded tetrahedral units in the melt is favoured [2].
A valence band emission study was performed on beamline ID26 to obtain temperature-dependent information of the valence states of liquid germanium well beyond the melting temperature using a Rowland spectrometer equipped with a spherical bent Si(931) crystal.
 |
|
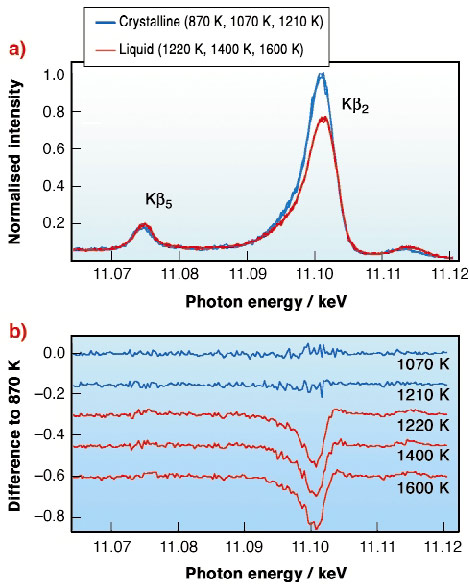
Fig. 8: a) Superposition of X-ray emission spectra taken at six different temperatures; b) Difference spectra in respect to the one taken at 870 K. |
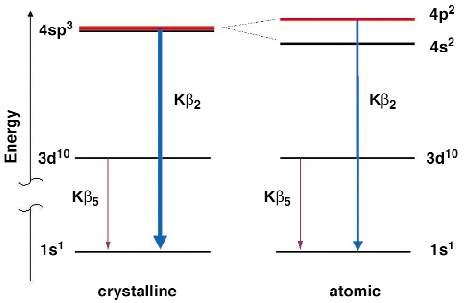
Figure 8a shows a series of spectra taken at six different sample temperatures, ranging from 870 K to 1600 K. All spectra are showing the Kß2 band which come from the dipole-allowed 4p-1s transition and the Kß5 line from the 3d-1s-quadrupolar transition. The most striking change upon melting is the decrease in intensity of the Kß2 emission (Figure 8b). The structure of crystalline germanium shows a local geometry similar to that found in simple tetrahedral molecules. This suggests that we should describe the bonding in terms of s and p orbitals and use the sp3-hybridisation as a first approximation to understand the electronic structure of crystalline and also of liquid germanium. The basic ideas are depicted schematically in Figure 9. The wave function of the four electrons participating in the tetrahedral covalent bond is a linear combination of one s- and three p- wave functions. Three electrons per atoms are contributing to the Kß2-emission. The isolated germanium atom has a 4s24p2 configuration and only two electrons are allowed to decay in the 1s core-hole. This argumentation strongly suggests that the intensity decrease of the Kß2 band upon melting is a direct consequence of the breaking of the covalent sp3-bonds in the crystalline solid. The melting process is accompanied by a relaxation of electrons with p-symmetry out of the sp3- bond to an energetically deeper lying s-state. Following this interpretation the intensity of the Kß2 band can be taken as a direct measure for the amount of covalently bonded tetrahedral units in the melt.
 |
|
Fig. 9: Schematic energy diagram showing the different contributions to the X-ray emission spectrum. |
Interestingly there is no further spectral evolution visible in the spectra by increasing the temperature. This indicates that the bonding properties around the absorbing atom in the liquid remain the same over the measured temperature range. Notably no gradual decrease in the concentration of covalent tetrahedral bonds in the melt can be detected, in contrast to the predictions of recent ab initio molecular-dynamics studies [3].
References
[1] V.M. Glazov et al., Liquid Semiconductors (Plenum, New York. 1969).
[2] N.W. Ashcroft, Nuovo Cimento D12, 597 (1990).
[3] R.V. Kulkarni et al., Physical Rev. B 55, 6896 (1997).
Author
T. Neisius.
ESRF



