- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2002
- Macromolecular Crystallography
- Protein and Virus Crystallography under High Hydrostatic Pressure
Protein and Virus Crystallography under High Hydrostatic Pressure
The potential of pressure as a thermodynamic variable in macromolecular crystallography remains largely unexplored although the pioneering work [1] on the structure of tetragonal crystals of hen egg-white lysozyme (tHEWL) compressed at 1 kbar in a polycrystalline beryllium cell was carried out as far back as 1987. This work was followed only recently by the publication of the 1.5 kbar structure of another small monomeric protein, sperm whale metmyoglobin, pressurised in a similar cell. A different device, a diamond anvil cell (DAC), has also been used to measure the compressibility of HEWL crystals although without follow-up data collection [2].
On beamline ID30, the combination of a DAC, ultra-short wavelength undulator radiation and a large imaging plate has allowed us to extend the field of high-pressure macromolecular crystallography in terms of both the accessible pressure range (increased by one order of magnitude with respect to the pressure range of beryllium cells), and the data quality obtainable. Experimental procedures were established and the first results obtained on crystals of tHEWL and Cu, Zn superoxide dismutase (SOD), a dimeric protein [3]. Compressibility measurements were achieved up to 8.2 kbar on tHEWL crystals and beyond 10 kbar for SOD crystals. Diffraction data from tHEWL crystals at 7 kbar were then collected and analysed demonstrating that high-pressure data can be of very similar quality (resolution 1.6 Å, completeness 0.93, multiplicity 7.2, Rmerge (I) 0.057) to that expected from experiments carried out at ambient pressure. tHEWL structures at normal pressure and 7 kbar were then refined in parallel at 1.6 Å resolution and a detailed comparison was made (example in Figure 13, article in preparation).
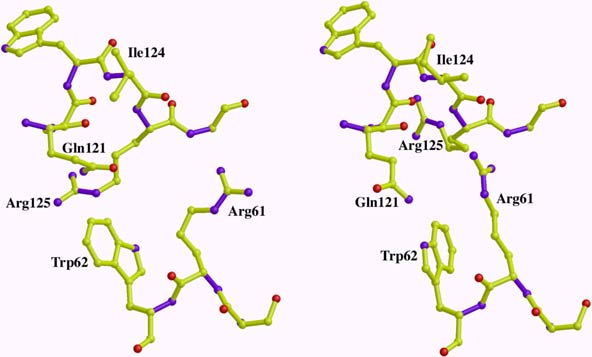 |
|
Fig. 13: A representation of a molecular contact region in lysozyme at ambient pressure (left) and at a pressure of 7 kbar (right). Several residues in the 7 kbar structure take new positions to accommodate the closer contacts induced by pressure between symmetry-related molecules in the crystal. |
We then applied the same methodology to crystals of cowpea mosaic virus (CPMV) particles, the first example of a crystallised macromolecular assembly studied at high pressure. CPMV is a member of the Comovirus family, a group of icosahedral plant viruses similar to mammalian Picornaviruses. The refined 2.8 Å crystal structure at atmospheric pressure was available [4]. Single oscillation images of a cubic crystal of CPMV were recorded at several pressures between 1 bar and 4.4 kbar at which point the crystal no longer diffracted. The initial crystal at 1 bar, 1.1 and 2.0 kbar was disordered and diffracted only to low resolution with the space group tentatively assigned as P23. At 3.3 kbar, however, the diffraction pattern extended to 2.6 Å with a high signal-to-noise ratio (Figure 14) and was indexed in space group I23. In this particular case, pressure induces a first order transition, with long-range three-dimensional order most likely reached through small rotations and translations of virus particles. The ordering effect of high pressure observed for CPMV was not unexpected, as an increase of pressure at constant temperature leads to a decrease of the entropy of the compressed system.
 |
|
Fig. 14: An oscillation image (0.3°) from an I23 form CPMV crystal at 3.3 kbar ( |
The type of disorder in CPMV at atmospheric pressure appears to be unusual, and further experiments on other systems are required. If it turns out that improvement of crystal, and hence data quality, under high pressure is a more general phenomenon, the consequences might be quite important for macromolecular crystallography. Besides this possible outcome, the demonstration that accurate structural information on macromolecular structures can be obtained at high pressure opens avenues such as the exploration of protein sub-states, the study of interactions between macromolecules and between subunits and the detection of the onset of pressure-induced denaturation.
References
[1] C.E. Kundrot and F.M. Richards, J. Mol. Biol. 193, 157 (1987).
[2] A. Katrusiak and Z. Dauter, Acta Cryst. D52, 607 (1996).
[3] R. Fourme, R. Kahn, M. Mezouar, E. Girard, C. Hoerentrup, T. Prangé and I. Ascone, J. Synchrotron Rad. 8, 1149 (2001).
[4] T. Lin, Z. Chen, R. Usha, C.V. Stauffacher, J.B. Dai, T. Schmidt and J.E Johnson, Virology 265(1), 20 (1999).
Principal Publication and Authors
R. Fourme (a), I. Ascone (b), R. Kahn (c), M. Mezouar (d), P. Bouvier (d), E. Girard (c), T. Lin (e) and J.E. Johnson (e), Structure 10, 1409 (2002).
(a) Synchrotron SOLEIL, Saint Aubin (France)
(b) LURE, Orsay (France)
(c) IBS, Grenoble (France)
(d) ESRF
(e) Scripps Research Institute, La Jolla (USA)



