- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2002
- X-ray Imaging
- Topographic and Quantitative Elemental Analysis of Human Central Nervous System Tissue
Topographic and Quantitative Elemental Analysis of Human Central Nervous System Tissue
Though well-characterised, the pathogenesis of neurodegenerative dissorders i.e. Parkinson's disease (PD) and amyotrophic lateral sclerosis (ALS) is still unknown. There is strong evidence that processes leading to degeneration and atrophy of nervous cells can be promoted by abnormal biochemical reaction catalysed by selected metal ions [1,2]. The main goal of this study is a better understanding of the role of metals in PD and ALS. The microbeam synchrotron X-ray fluorescence (m-SXRF) technique was used to obtain topographic and quantitative analysis of selected elements in central nervous system (CNS) tissue from PD, ALS and control cases. Two areas of CNS, i.e. substantia nigra (SN) of brain and thoracic spinal cord were sampled. Nonstained, cryomicrotome slices were investigated.
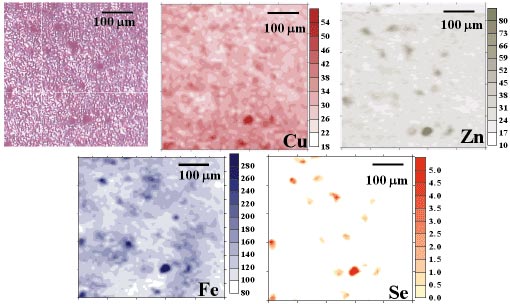
The measurements were carried out on beamline ID22. The polychromatic pink beam mode excitation was used at a final beam spot of 5 µm x 2 µm (horizontal x vertical). The elements such P, S, Cl, K, Ca, Fe, Cu, Zn, Se, Br, Rb and Sr were identified in CNS tissue. Two-dimensional maps of elemental distribution were compared with the microscopic view of the histopathological sections. The results showed that significantly higher intensities of selected elements in m-SXRF images correspond to the position of the neurons in the tissue slices. Particularly, in the control group, neurons of substantia nigra revealed higher accumulation of S, Cl, K, Ca, Fe, Zn, Se and Rb when compared to the surrounding area. Figure 107 shows the distribution of Cu, Zn, Fe and Se together with the optical image of the scanned area of the control tissue.
 |
|
Fig. 107: Substantia nigra the control case: (a) microscopic view of scanned area of the tissue; and (b) distribution of selected elements. The neurons are seen on the optical image as the dark points. The values on the scales represent intensities in relative units. |
The elemental maps obtained from the tissue slices of spinal cord show, in the control case, a correlation between neuron bodies positions and increased content of S, Cl, K, Ca, Zn, Br and Rb. Such correlation is not observed for Fe and Cu, whereas P is accumulated to a lower degree inside the perikarial part of the neuron. The topographic results obtained for ALS tissue slices of spinal cord are in good agreement with those observed for the control group excluding S.
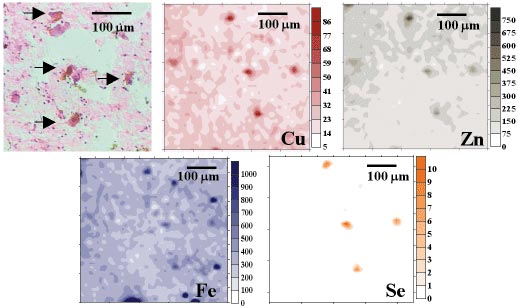
The distribution of selected elements in PD tissue is presented in Figure 108. The quantitative analysis indicated that SN neurons of the PD samples show a strong accumulation of Cu and Fe and a significantly higher accumulation of S, Ca, Zn, Se and Br in comparison with the control case. For the same part of the brain, an increased content of Cl, Ca, Zn, and Br was noticed for the ALS case. The quantitative analysis reveals a decreased level of Zn in the degenerated motor neuron of spinal cord in the ALS cases studied.
 |
|
Fig. 108: Substantia nigra the PD case: (a) microscopic view of scanned area of the tissue; and (b) distribution of selected elements. The arrows show neuron localisation. The values on the scales represent intensities in relative units. |
The present study indicated that µ-SXRF is a very powerful tool for the study of abnormalities in elemental concentrations, in thin CNS tissue sections at the single cell level. These abnormalities may promote biochemical reactions leading to pathological changes in CNS tissue especially to the degeneration and atrophy of neurons. Our results are in good agreement with existing hypotheses concerning the neurodegenerative role of trace elements, which are based on in vitro investigations or experiments on animals. These results should help us gain some information on metal function in the central nervous system tissue for neurological disorders. Particularly interesting is the perspective of using this tool for the investigation of chemical states of metallic elements in tissues from sites of degeneration.
References
[1] L.M. Sayre, G. Perry, M.A. Smith, Current opinion in Chemical Biology 3, 220-225, (1999).
[2] J.S. Bains, C.A. Shaw. Brain Res. Rev. 25, 335-358, (1997).
Authors
M. Lankosz (a), M. Szczerbowska-Boruchowska (a), J. Ostachowicz (a), D. Adamek (b), A. Krygowska-Wajs (b), B. Tomik (b), S. Bohic (c), A. Simionovici (c).
(a) Faculty of Physics and Nuclear Techniques, University of Mining and Metallurgy, Krakow (Poland)
(b) Institute of Neurology, Collegium Medicum, Jagiellonian University, Krakow (Poland)
(c) ESRF



