- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2001
- Surfaces, Interfaces and Nano-science
- The Adsorption of Carbon Monoxide on Ni(110) Above Atmospheric Pressure Investigated with Surface X-ray Diffraction
The Adsorption of Carbon Monoxide on Ni(110) Above Atmospheric Pressure Investigated with Surface X-ray Diffraction
Since the discovery in the nineteenth century that some gas molecules adsorbed onto a metal surface are readily converted into other molecules, heterogeneous catalysis has achieved tremendous technological, environmental, and commercial importance. Understanding the gas-metal interaction by determining the structures of adsorbed molecules is a primary goal of modern surface science. The adsorbate geometry of gases on metal surfaces has thus been determined and catalogued for more than one thousand systems under the extreme vacuum conditions of 10-8 - 10-14 bar where appropriate techniques were available. As most of the commonly employed techniques for surface structure determination involve electrons (e.g. electron diffraction, photoemission) they are not suitable for investigating gas/solid interfaces at pressures near atmospheric. Consequently, an essential question remained unanswered: "Are the known vacuum structures also the relevant structures present under real catalytic conditions near 1 bar?"
Here, we answer that question affirmatively (at room temperature) and negatively (at elevated temperature) for the archetypal case of CO over Ni(110). Based on X-ray diffraction measurements, the CO/Ni(110) structure was determined in situ from 10-10 to 2.3 bar CO at 25°C. Interestingly, the vacuum structure persisted unchanged over ten orders of magnitude of pressure. A subsequent warming to ~ 130°C at 2.3 bar then caused a massive restructuring of the Ni surface consisting of the development of microfacets with (111) orientation and surface strain probably due to carbon dissolution. These results confirm the relevance of vacuum studies to catalysis and offer a glimpse at the complexity of elevated-pressure surface chemistry.
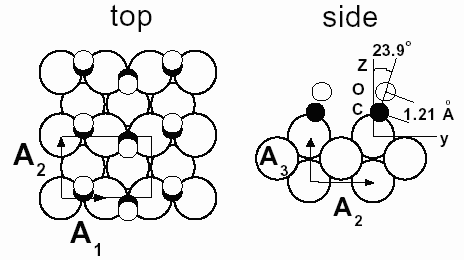 |
Fig. 64: Top and side views of the CO/Ni(110) 2x1 structure. A1, A2 and A3 are the lattice vectors used to describe the crystal lattice. A1 = A3 = a0/ |
Nickel catalysts are used most frequently to produce methane from carbon monoxide and hydrogen, at temperatures in the range 150 - 400°C and pressures around 1 bar. In vacuum, adsorbed CO on Ni(110) forms a 2x1 structure consisting of an ordered zigzag arrangement of tilted molecules on short-bridge sites of the substrate as depicted in Figure 64. The structural parameters obtained from the most recent LEED (low energy electron diffraction) study [1] are given in Table 1.
 |
Table 1: Structural parameters of CO/Ni(110)(1x2). |
The experiments reported here were performed at the Surface Diffraction Beamline ID3 with a specially designed ultra-high-vacuum (UHV)/high pressure chamber. We collected two independent sets of crystallographic data in situ after exposing a well prepared Ni surface to 10-10 bar and 2.3 bar of CO. Figure 65 shows the results. The top panel gives the structure factors of six diffraction rods [i.e IH,K (L)]. Four have integer-valued H and K corresponding to the periodicity of the substrate lattice and two rods have fractional index arising from the CO 2x1 periodicity. The bottom panel displays the structure factors of the fractional reflections at L 0. Inspection of the figure shows immediately that the vacuum and high-pressure data sets are virtually identical and thus demonstrates that both structures are the same. Crystallographic analysis of the high pressure data through a least squares minimisation routine results in the continuous red curve in the figure. The best fit model resulted in the structural parameters shown in Table 1. The agreement with the results of Zhao et al. [1] is excellent, and additionally, our analysis reveals a slight expansion of the Ni planes, which had not been detected. The table also shows the results of the analysis of the low-pressure data, which coincide with those at high pressure.
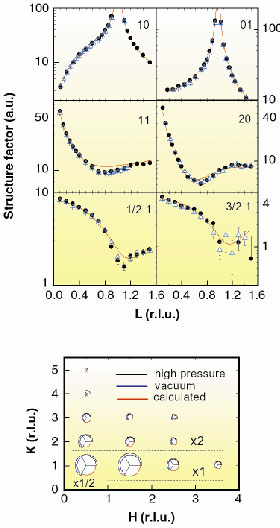 |
Fig. 65: Crystallographic structure factors of CO/Ni(110)(1x2) at room temperature. Upper panel: Black circles are measured structure factors from four integer rods [(H, K) = (1,0), (0,1), (1,1) and (2,0)] and two fractional order rods [(H,K)= (1/2,1), (3/2, 1)] in 2.3 bars of CO; Blue open triangles: structure factors measured in 10-10 bars of CO; Red solid lines are calculated structure factors from our best fit to the high-pressure data. In the integer rods, the diverging values of the structure factors at some integer values of L correspond to the bulk Bragg reflections of the Ni crystal. Lower panel: In-plane fractional order structure factors measured at L = 0.1. The radius of the black 120° sectors are proportional to the structure factors of the reflections from the structure in 2.3 bar of CO. The error bars are indicated by the two radii. The blue sectors represent the structure factors for the vacuum structure and the red ones are calculated from the fit to the high-pressure data. |
From the previous experiments we can conclude unambiguously that the equilibrium structure at room temperature of CO on Ni(110) at 2.3 bars of CO ambient pressure is the same as that obtained under UHV conditions by dosing the Ni(110) surface at saturation with 10-10 bar of CO. To our knowledge this is the first chemisorbed structure determined in detail near atmospheric pressure.
Reference
[1] C. Zhao , M.A. Passler , Surf. Sci., 320, 1-6 (1994).
Principal Publication and Authors
K.F. Peters, C.J. Walker, P. Steadman, O. Robach, H. Isern and S. Ferrer, Phys. Rev. Lett. 86, 5325 (2001).
ESRF



