- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2001
- Magnetism and Electronic Properties of Solid
- Charge Ordering in Magnetite Below the Verwey Transition
Charge Ordering in Magnetite Below the Verwey Transition
Magnetite (Fe3O4) is the eponymous magnetic material, whose properties were first recorded by Greek writers in ~800 B.C. At ambient temperatures magnetite is ferrimagnetic and electrically conducting, with a cubic inverse spinel crystal structure. Below the 122 K Verwey transition [1], the conductivity falls by a factor of ~100 and a small structural distortion is observed. This transition has been assumed to be the result of Fe2+/Fe3+ charge ordering, although a detailed picture of the low temperature phase has yet to emerge.
In order to further characterise the low temperature structure we have refined the crystal structure of magnetite at 90 K using very highly resolved powder X-ray (BM16) and neutron diffraction data (HRPD, ISIS). The compression of the three dimensional reciprocal space into one dimension in the powder diffraction pattern is compensated by avoiding many of the technical difficulties associated with single crystal experiments for this sample, which include severe twinning, extinction and multiple scattering effects.
The refined model represents an averaging of the true low temperature structure, as a smaller unit cell and non-crystallographic symmetry constraints were required for convergence of the refinement. Despite these limitations the model accounts well for the intensities of the vast majority of the superstructure reflections, with only very few weak reflections remaining unindexed. The model is similar to previous results [2] but shows that two of the four unique octahedral iron sites have, on average, significantly longer Fe-O distances than the other two. This provides direct crystallographic evidence for at least partial long-range charge ordering in magnetite, with an apparent difference of 0.2e- between the average charges at the two sets of sites. Figure 103 shows a small region of the fit to the X-ray data, plotted on a logarithmic scale so that the weak superstructure peaks can be seen. Data collected above the Verwey transition, at 130 K, are also shown displaced above the fit for comparison.
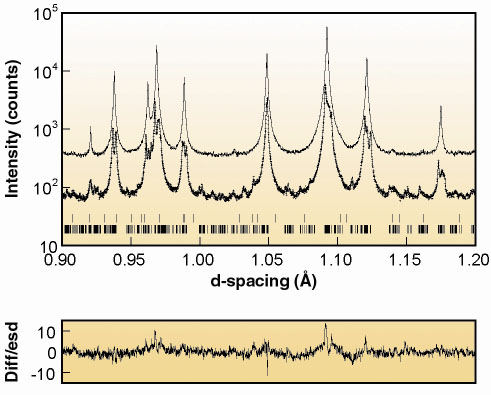 |
Fig. 103: Profile fit to BM16 powder diffraction data for Fe3O4 Upper and lower tick marks refer to Fe2O3 (0.8 wt.%) and Fe2O3 respectively. |
It is possible to account for the pattern of displacements of the ions in terms of a [001] charge density wave in the reduced unit cell that was used for refinement. Alternatively, charge-ordered models can be proposed by generating all possible charge-ordering schemes consistent with the larger unit cell and comparing them to the refined model in the smaller unit cell. Figure 104 shows the model having the lowest electrostatic repulsion energy while remaining consistent with the data. While our results do not distinguish between these alternatives, it is clear that none of the charge-ordering schemes that give the minimal electrostatic repulsion are consistent with the refined model. Thus, charge-ordering schemes consistent with our refinement do not meet the widely accepted Anderson criterion of minimum electrostatic repulsion. This indicates that the low temperature structure of magnetite is a compromise between electrostatic repulsion and structural distortions and suggests that the transition is driven by an [001] electronic instability, which opens a band gap through a charge density wave mechanism.
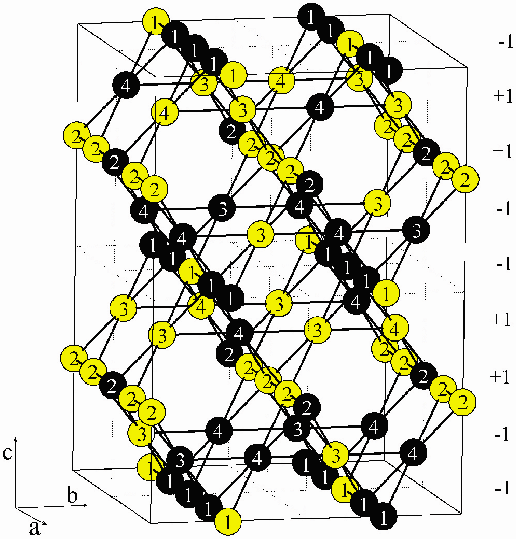 |
Fig. 104: A model charge-ordering scheme for the low temperature phase of Fe3O4. Labels 1-4 refer to distinct crystallographic sites with black and yellow circles indicating Fe2+ and Fe3+, respectively. |
References
[1] E.J.W. Verwey, Nature, 144, 327-328 (1939).
[2] M. Iizumi, T.F. Koetzle, G. Shirane, S. Chikazumi, M. Matsui and S. Todo, Acta. Cryst, B38, 2121-2133 (1982).
Principal Publication and Authors
J.P. Wright (a), J.P. Attfield (b) and P.G. Radaelli (c), Phys. Rev. Lett., 87, 266401 (2001).
(a) ESRF
(b) Chemistry Department, University of Cambridge (UK)
(c) ISIS Facility, Rutherford Appleton Laboratories, Chilton (UK)



