- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2001
- Chemistry
- BioXAS at Room Temperature
BioXAS at Room Temperature
Understanding the relationship between structure and function is one of the main interests in the molecular biosciences. X-ray absorption spectroscopy on biological samples (BioXAS) is highly useful for characterisation of the structure of protein-bound metal complexes. Progress in the performance of synchrotron radiation sources and of experimental stations dedicated to the study of ultra-dilute biological samples has made it possible to carry out new types of BioXAS experiments, which were impracticable in the past [1]. The first steps towards atomic-resolution studies on biological catalysis at quasi-physiological conditions (non-crystalline samples, room temperature) are reported for the manganese complex of photosynthesis.
Essentially all molecular oxygen (O2) in the Earth's atmosphere has been produced by light-driven water oxidation at the tetra-manganese complex of photosystem II (PSII). The underlying chemistry seems to be unique and is of potential technological interest. Structural information has become accessible by X-ray absorption spectroscopy at the Mn K-edge carried out at liquid-helium temperatures [2]. Previously detailed information on structure and oxidation state of the PSII manganese complex at room temperature was not available, so we investigated the tetra-manganese complex at 290 K by XAS on PSII membrane particles.
 |
| Fig. 39: Sample simultaneously exposed to the X-ray beam and to visible light which drives photosynthetic water oxidation. In the photograph, the X-ray beam enters from the upper right edge and passes through an ion chamber before it hits the PSII sample (marked by the yellow arrow). The X-ray fluorescence is detected at right angles by a photodiode (the round device behind the sample). The temperature of the sample is controlled by a stream of nitrogen delivered by the metallic pipe in the left part of the photograph. To drive the photoenzyme through its catalytic cycle, samples are illuminated with a sequence of nanosecond flashes of green laser-light. |
The experiments were carried out at ID26, where simultaneous scanning of the monochromator and the undulator gap facilitates rapid measurement of fluorescence-detected X-ray absorption spectra on ultra-dilute samples (Figures 39 and 40). Owing to the rapid-scan capabilities of ID26, i.e. collection of EXAFS spectra in less than 10 seconds, any significant influence of X-ray radiation damage is avoidable, and advanced room-temperature BioXAS investigations on the PSII in its native membrane environment become feasible. At 290 K, as well as at 18 K, the manganese complex in its dark-stable (S1) state is apparently a Mn(III)2Mn(IV)2 complex, comprising two di-µ2-oxo bridged binuclear manganese units characterised by the same Mn-Mn distance of 2.71-2.72 Å. Most likely, the manganese oxidation states and the protonation state of the bridging oxides are fully temperature independent. Remarkably, at room temperature manganese-ligand distances of 3.10 Å and 3.65 Å are clearly discernible in the EXAFS spectra. The type of bridging assumed to result in Mn-Mn or Mn-Ca distances around 3.1 Å is, possibly, temperature dependent as suggested by distance lengthening by 0.13 Å on cooling.
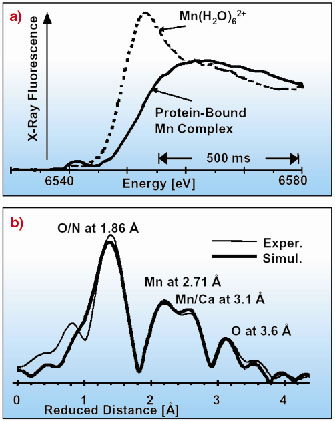 |
Fig. 40: a) Edge spectra of the intact PSII manganese complex (solid line) and of the Mn2+ ions (broken line) formed after 150 s of X-ray exposure of the biological sample at 290 K. The displayed spectrum was collected in 1 s; averaging of scans was not employed. b) Fourier-transformed k3-weighted EXAFS spectra. Twelve EXAFS scans of 10 s duration had been averaged. |
Room-temperature XAS investigations open a new way to gain insights into the structure-function relationships. Structural investigations under defined (with respect to pH, redox equilibria, etc.) and 'almost-native' conditions become viable. The ambiguity with respect to the relevance of low-temperature results is avoidable. Furthermore, the capability to investigate the metalloenzyme at its working temperature is the prerequisite for time-resolved XAS investigations aiming at observation of structural changes in real time (without using a freeze-quench approach). Recently, using laser-flash excitation of a sample exposed to the X-ray beam on ID26 (Figure 39), we succeeded in observing directly the advancement in the catalytic cycle by XAS at room temperature (unpublished results).
References
[1] H. Dau and M. Haumann, J. Synchrotron Rad. (submitted).
[2] H. Dau, L. Iuzzolino and J. Dittmer, Biochim. Biophys. Acta 1503, 24-39 (2001).
Principal Publication and Authors
C. Meinke (a), V.A. Solé (b), T. Neisius (b), P. Pospisil (c), M. Haumann (d) and H. Dau (d), Biochemistry 39, 7033-7040 (2000).
(a) FB Biologie, Philipps-Universität Marburg (Germany)
(b) ESRF
(c) Harvard University, Cambridge (USA)
(d) FB Physik, Freie Universität Berlin (Germany)



