- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2000
- Surfaces and Interfaces
- Compression Versus Expansion on Ionic Crystal Surfaces: ADP{101}
Compression Versus Expansion on Ionic Crystal Surfaces: ADP{101}
The atomic structure at the solid/liquid interface will in general control the growth of a crystal in solution. The technique of surface X-ray diffraction (SXRD) allows us, for the first time, to 'look' in situ at such an interface and thus to address the role of surface relaxations, reconstructions, impurities and also to distinguish between various crystal terminations. Such structural details are typically not taken into account in the current theories that predict the growth shape of a crystal [1]. We are particularly interested in the surface structure of ionic crystals such as ADP (ammonium- dihydrogen phosphate NH4H2PO4), a crystal of the KDP family, because of their use in optical switching and frequency conversion in laser applications and because of their dominant role in fundamental studies in the solution growth field.
The Hartman-Perdok theory correctly predicts that the pyramidal {101} and the prismatic {100} faces of ADP are present during solution growth. The ADP{101} face can, in principle, terminate with an [NH4]+ (ammonium) or [PO4]3 (phosphate) layer, or both. AFM (atomic force microscopy) measurements have shown that the height of the steps on the {101} faces of ADP corresponds to the thickness of double layers, which means that only one termination is present.
SXRD measurements were performed at the ID32 beamline and the data consist of a set of crystal-truncation rods. Figure 49 shows measured structure factor amplitudes along the (10) CTR for ADP{101}. Our data show that the surface layer of ADP{101} is [NH4]+ terminated in the growth solution. The data is of sufficient quality to determine the relaxations of the outermost layers as well. We find that ammonium is relaxed inwards by 0.32 ± 0.13 Å and phosphate is relaxed 0.25 ± 0.11 Å outwards.
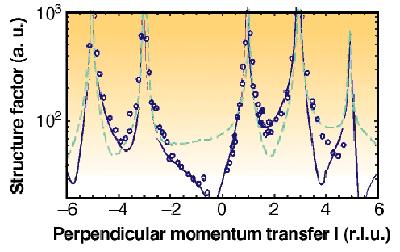 |
Fig. 49: Structure factor amplitudes along (10) crystal truncation rod, for ADP(101). The solid line is the best fit starting from a [NH 4] + terminated surface with relaxations included. Dashed line is the calculation for a H 2[PO 4] terminated one.
|
It is interesting to compare our results for ADP with the isomorphous KDP surface [2]. In both cases, the crystal terminates with the positive layer. However, while for ADP the top layer moves inward, for KDP it relaxes outwards. For both systems, the second layer relaxes outwards. A second difference is that the relaxations are much larger for ADP than for KDP. It seems likely that in ADP the hydrogen bonding of the top [NH4+] layer with the liquid layer plays a role. This mechanism is absent in the case of KDP. The second layer consists in both cases of [H2PO4]- and these show similar behaviour.
References
[1] P. Bennema, Growth and Morphology of Crystals, Chap.7 in Handbook of Crystal Growth, vol.1a (North Holland, Amsterdam, 1993).
[2] S.A. de Vries, P. Goedtkindt, S.L. Bennet, W.J. Huisman, M.J. Zwanenburg, D.M. Smilgies, J.J. De Yoreo, W.J.P. van Enckevort, P. Bennema and E.Vlieg, Phys. Rev. Lett. 80, 2229 (1998).
Authors
J. Arsic, M. Reedijk, A. J. R. Sweegers and E. Vlieg.
University of Nijmegen (The Netherlands)



