- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2000
- Life Sciences
- The Light-driven Chloride Pump Halorhodopsin Equivalent Mechanisms for Proton and Chloride Transport?
The Light-driven Chloride Pump Halorhodopsin Equivalent Mechanisms for Proton and Chloride Transport?
Halide transport through biological membranes imposes structural restraints on membrane transporters that are different from those in the transport of protons or cations. Apart from well-known examples of chloride transporters such as the CFTR chloride channel or the band 3 protein, halorhodopsin (HR) is the only representative of a light-driven chloride pump. This heptahelical membrane protein is an archaeal rhodopsin that occurs ubiquitously in Haloarchaea. After light absorption at 578 nm by an isomerisable retinal-Schiff base chromophore, chloride is pumped through the lipid bilayer in a subsequent series of spectroscopically distinguishable intermediates.
Three-dimensional crystals of halorhodopsin were grown in a cubic lipidic phase using conditions that closely mimicked the hypersaline conditions of the haloarchaeal cytosol. The structure of this anion pump was determined at 1.8 Å resolution using high-quality data collected at beamline ID14-3 [1]. The layer-like packing of halorhodopsin monomers in these 3D-crystals closely matches the organisation of purple membranes which are natural 2D-crystals formed by the homologous proton pump bacteriorhodopsin. In a similar manner to bacteriorhodopsin, halorhodopsin assembles into trimers around a central lipidic patch, but adopts a different quaternary arrangement [2]. Next to the protonated Schiff base between Lys 242 and the isomerisable retinal chromophore, a single chloride ion, CL501, occupies the transport site (Figure 3). The location of CL501 precisely coincides with the position of the primary proton-accepting oxygen of D85 in bacteriorhodopsin (BR) which indicates that the electrostatics are highly conserved in the transport sites of HR and BR despite the different transport specificities. The structure of HR now suggests a simple mode of ion dragging across the PSB such that chloride and proton translocation modes are mechanistically equivalent in archaeal rhodopsins.
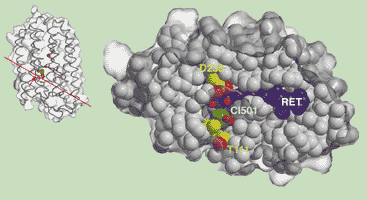 |
Fig. 3: The chloride-binding site in halorhodopsin. CL501 (green sphere) occupies the transport site of HR, with an attached cluster of three water molecules (small, red spheres). The retinal chromophore (RET, blue) contacts CL501 with its Schiff base nitrogen atom. Photoisomerisation of the retinal and subsequent structural changes in the protein portion, which are currently being studied using cryo-trapped crystals, cause the translocation of CL501 towards the other side of the retinal and final release towards the cytosol.
|
References
[1] M. Kolbe, H. Besir, L.-O. Essen and D. Oesterhelt, Science, 288, 1390-1396 (2000).
[2] L.-O. Essen, R. Siegert, W.D. Lehmann and D. Oesterhelt, Proc. Natl. Acad. Sci. USA, 95, 11673-11678 (1998).
Authors
L.-O. Essen, M. Kolbe, H. Besir and D. Oesterhelt.
Max-Planck-Institute for Biochemistry, Martinsried (Germany)



